MannKind’s inhaled insulin drug proves hard for diabetics to get

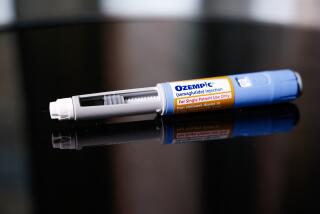
The inhaled insulin drug Afrezza is administered through a device about the size of a small whistle.
It took 10 years and $1 billion for MannKind Corp. in Valencia to get its breakthrough inhaled-insulin drug approved by U.S. regulators.
Now comes the hard part: getting people with diabetes to use it.
If Cynthia Goldstein’s experience is any indication, it’s not going to be easy.
The West Los Angeles woman said she encountered many obstacles before she got her hands on the new drug: first from a doctor reluctant to prescribe it, then from Medicare and finally, her pharmacy.
“If I weren’t the world’s most persistent person, I would have given up a long time ago,” Goldstein said. “They have a great product, and they can’t get it into the hands of consumers because of the rest of the process. There’s just so many roadblocks.”
The new drug, Afrezza, was expected to be the latest breakthrough for entrepreneur Alfred Mann, the 89-year-old founder and chairman of MannKind, who scored previous successes with pacemakers, insulin pumps and hearing implants.
But early sales have been a disappointment — just $1.1 million in its first two months on the market, not a good start for a drug that some analysts expected to top $1 billion in annual sales.
Mann says it’s far too early to worry, and he boasts that Afrezza will become one of the top-selling drugs in history.
To reach that goal, MannKind and its marketing partner, French pharmaceutical company Sanofi, must overcome several hurdles.
Afrezza faces extensive competition from existing drugs, including oral medication and injections that are getting easier to administer. “Their most immediate competition is the fast-acting, injectible insulins, which people are doing pretty well with,” said Dr. Mark O. Goodarzi, director of the endocrinology division at Cedars-Sinai Medical Center in Los Angeles.
“I personally haven’t prescribed it yet,” he said. “None of my patients have been so unhappy about injections that they want to switch to the inhaler.”
Another obstacle is a U.S. Food and Drug Administration requirement that patients pass lung-function tests before they can use the drug. The FDA required MannKind to include a warning that the inhaled insulin could cause health complications for people with lung disease. Smokers should not use the drug, the FDA says.
Health insurers have caused further delays, with some requiring special authorization before they’ll pay for the new drug. It’s likely that Afrezza will cost more than injected insulin, but pricing depends on many factors, including insurance coverage and dosage.
Stefan Schwarz, head of Sanofi’s U.S. marketing team for Afrezza, said the wholesale price for 20 units a day of Afrezza is $9.29, compared with $5.23 for its injected insulin, Apidra. Sanofi is offering coupons on its website to help patients cover co-pays.
The good news for MannKind and Sanofi is that most patients who try the new insulin like it, both for its ease of use and effectiveness.
“The nice thing about Afrezza is it works instantly for me,” said Goldstein, 66, who has had Type 1 diabetes for more than 15 years. “I take my puff after I’ve been eating awhile, and my blood sugar never goes really high.”
Afrezza is a powder that is administered through a device about the size of a small whistle.
The fast-acting insulin is intended to be used at mealtime, a quick puff replacing an injection. It is the only drug that MannKind has taken to market and the key to the future of the company, which employs about 25 people at its Valencia headquarters and 250 at its manufacturing plant in Connecticut.
Matt Pfeffer, MannKind’s chief financial officer, said Afrezza’s success depends on getting doctors and consumers to try it.
“We only have to get them to try it once, because if the pattern holds, they’re going to like it a lot,” Pfeffer said.
Dr. Alan Marcus, a diabetes doctor in Laguna Hills, said he has followed the drug’s development for more than a decade.
In three months, he’s already prescribed the drug to about 30 patients. “They love it,” he said. “It allows them to control their glucose ... in a manner that doesn’t stigmatize. If you’re out at a restaurant, the last thing you want to do is go to the bathroom for an injection or stick a needle through your clothing.”
But unless something is done to make it easier to get the drug into patients’ hands, doctors will probably be reluctant to prescribe it, Marcus said.
“It’s not an easy prescription,” he said. “They have to go through testing. Then you have to take a moment to demonstrate it. Then you have to deal with the insurance companies. Then you have to deal with the pharmacists, because most of the pharmacies don’t have it in stock. This is a process that takes a lot of time, it takes a lot of effort.”
Creating an effective drug is no guarantee it will be come a blockbuster, especially when there are several other medicines on the market. Pharmaceutical companies are also not known for their patience with drugs that start slowly.
“Drug companies very often abandon things if they don’t come out of the gate like trailblazers,” said David Maris, a pharmaceutical industry analyst with BMO Capital Markets. “It’s like the movie industry: If it doesn’t have a big box office the first weekend, the movie studios back away from advertising and pushing it.”
Blockbuster drugs are rare. Gilead Sciences Inc.’s hepatitis C drug Sovaldi generated $10.3 billion of sales in 2014, making it one of the top-selling drugs in the world in its first year on the market. But Sovaldi cured a disease, while Afrezza is one option in a crowded field of diabetes treatments.
“The single biggest factor will be Sanofi’s commitment to marketing the product,” Maris said. “Getting people aware of things and getting them to prescribe it and stick with it is expensive and it takes time.”
To this point, the biggest marketing push has come from Afrezza users, who have taken to social media sites such as Twitter and Reddit to spread the word.
Sam Finta, a diabetes patient from Atlanta, said he’s shared his experiences with the drug with people around the world through his Twitter account: @afrezzauser. Launched in December, the account now has about 1,500 followers.
Finta said the drug works much more quickly than injected insulin, without the concern of reducing blood sugar to an unsafe level.
“It kicked-in in 15 minutes and was out of my system in an hour,” Finta said, adding that it’s been “life-changing.”
Despite the Internet buzz, J.P. Morgan analyst Cory Kasimov issued a brutal view of MannKind in a May 11 report. He said he now expects Afrezza sales to reach just $500 million by 2019 — 50% less than his earlier estimate — largely because the required lung testing is turning off physicians and patients.
He said lung-testing requirements were a big reason that Pfizer’s inhaled insulin Exubera was discontinued in 2007 because of poor sales after just a year on the market.
“We are increasingly skeptical that [Afrezza] will gain material traction any time soon, which could put the balance sheet into an increasingly precarious position,” Kasimov said.
Shaunak Deepak, an analyst with Jefferies in New York, is much more optimistic. In a May 15 research note, Deepak said demand for the drug is likely to rise as more doctors become familiar with it. Those doctors who are aware of the drug are prescribing it “more frequently than we had expected, especially among Type 1 diabetics,” Deepak said.
Investors pummeled MannKind’s stock for much of this year, but the stock soared 33% in the first three days of trading after Deepak’s report. The stock closed Tuesday at $5.21, down 50% in the last year.
Mann, the founder and executive chairman of MannKind, said it’s far too early for concern.
“It’s going to take a couple of quarters before you really see the numbers, but the results are so good that I can’t imagine it being a long-term problem,” he said.
Comparisons to the failed Pfizer drug are off-base, he said. Afrezza acts faster than Exubera did and it is much easier to use. The Pfizer drug was inhaled through a device about the size of a can of tennis balls, which made it awkward, Mann said.
The task of building market share for Afrezza falls to Schwarz, the Sanofi marketing leader for Afrezza.
He said Sanofi is taking a two-pronged approach: first educating physicians about the drug, then launching an advertising campaign — planned for later this year. Schwarz would not disclose the size of the sales team that’s marketing Afrezza, but he said sales reps are in all 50 states. Sanofi has about 17,000 employees in the United States.
“We do cover the entire nation,” he said. “There are no white spots. This is too important.”
Twitter: @spfeifer22
More to Read
Inside the business of entertainment
The Wide Shot brings you news, analysis and insights on everything from streaming wars to production — and what it all means for the future.
You may occasionally receive promotional content from the Los Angeles Times.