Opinion: DEA makes Vicodin harder to obtain legally, but that’s half the job
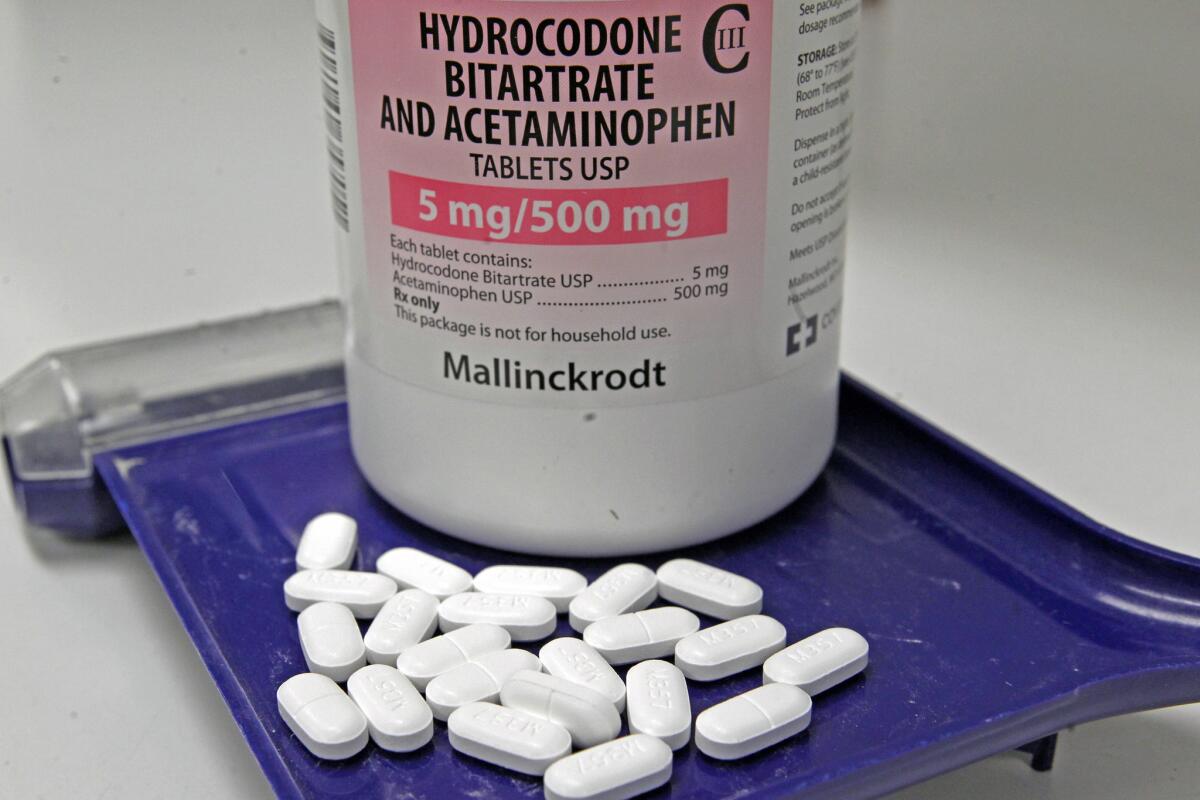
The Drug Enforcement Administration has spent the last decade considering whether to make it harder to obtain the most popular type of narcotic painkiller: “hydrocodone combination” products, such as Vicodin and Lortab. And now it will. But the new restrictions may not have much effect on stopping the abuse of those products until state officials make it harder for addicts to get around the rules.
On Friday, the DEA published a final rule that reclassifies hydrocodone combination products as Schedule II drugs, which is the most restrictive category for controlled substances that have medical uses. As such, they will be available only in non-refillable prescriptions that provide no more than a 90-day supply of the medication.
The change will certainly make it tougher for people to obtain these pills. Some pharmacies don’t dispense Schedule II drugs, and some states (though not California) bar nurse practitioners and physicians’ assistants from prescribing them. The ban on refills, meanwhile, could nip addictions in the bud by having doctors see the patients taking these medications more frequently.
The missing piece, though, is a mechanism to stop those who are determined to obtain a limitless supply of the drug -- that is, addicts -- from obtaining prescriptions from insufficiently cautious physicians or unscrupulous doctors willing to feed their habits. The Times’ Lisa Girion and Scott Glover wrote a damning series of stories about the latter, revealing that a few dozen doctors each had at least three patients who had fatally overdosed on the drugs the physicians had prescribed. The series also noted how ineffective the state’s medical board had been in policing its ranks.
California actually is one of the few states that has laid the foundation for solving both of these problems. Its CURES database and prescription drug monitoring program collects data from pharmacies about all the controlled substances that are dispensed. Doctors registered with the program can check the database to see whether a patient seeking pain medication has received it already, and if so, when and how much.
The operative word here is “can” check the database, not “must.” Nothing compels anyone -- prescribers or dispensers -- to look at CURES before hooking someone up with a 90-day supply of opiod. The Legislature considered a bill that would have required the prescription-writer and the dispenser to check CURES before supplying a patient with certain controlled substances for the first time. But the California Medical Assn. lobbied hard against the mandate, arguing that CURES needed far more resources to handle that kind of use. The final version of the measure provides more resources to increase the capacity of CURES and requires prescribers and pharmacists to sign up for access to it, but doesn’t require them to use it.
In a recent interview, Dr. Richard E. Thorp, president of the state medical association, insisted that physicians want to use CURES because it helps them identify patients who are “doctor shopping.” But without a mandate to use it, CURES won’t be an effective safeguard against addicts taking advantage of less diligent prescribers or pill-pushers such as the ones Girion and Glover chronicled.
Proposition 46 on the November ballot includes just such a mandate on doctors prescribing or pharmacists dispensing Schedule II, III and IV drugs (that is, drugs with at least some potential for abuse) to a patient for the first time. But the proposition wouldn’t delay the mandate, even though CURES won’t be able to handle the extra demand for months.
The medical association is one of the groups opposing Proposition 46, and not just because of the mandate to use CURES. The measure would also require random drug testing for physicians with hospital admitting privileges and would more than quadruple the cap on damages for pain and suffering caused by medical malpractice. The current cap, $250,000, was set when California overhauled its medical malpractice laws in 1975 and hasn’t been raised since.
For the record, The Times’ editorial board argued against reclassifying hydrocodone compound drugs in January 2013, saying a more effective approach would be to make better use of CURES. “The benefits of having hydrocodone more readily available to those who need it appear for now to outweigh the toll of its misuse,” we wrote. “Though that picture might change if hydrocodone abuse continues to increase, for now the FDA should continue to classify it as a less tightly restricted Schedule III drug.”
Schedule II drugs already include the pure form of hydrocodone. But medications that mix hydrocodone with other, less popular therapeutic substances were classified as Schedule III drugs, meaning that doctors could issue refillable prescriptions that provided half a year’s worth of pills -- assuming they were taken in the prescribed dosage.
The DEA first considered reclassifying hydrocodone combination products in response to a petition in 2004, but the Department of Health and Human Services effectively vetoed the shift in 2008. Rising opiod abuse and addictions led Congress to tell the administration to reconsider, and in 2013 an interagency drug safety panel and HHS recommended that the medication be reclassified. HHS offered three reasons: overmedicated individuals posed a threat to themselves and others, a significant amount of the drugs were being diverted to people who hadn’t been prescribed them, and people were taking them without the advice of a physician.
Follow Healey’s intermittent Twitter feed: @jcahealey
More to Read
A cure for the common opinion
Get thought-provoking perspectives with our weekly newsletter.
You may occasionally receive promotional content from the Los Angeles Times.