FDA panel recommends continued Avandia sales, with restrictions
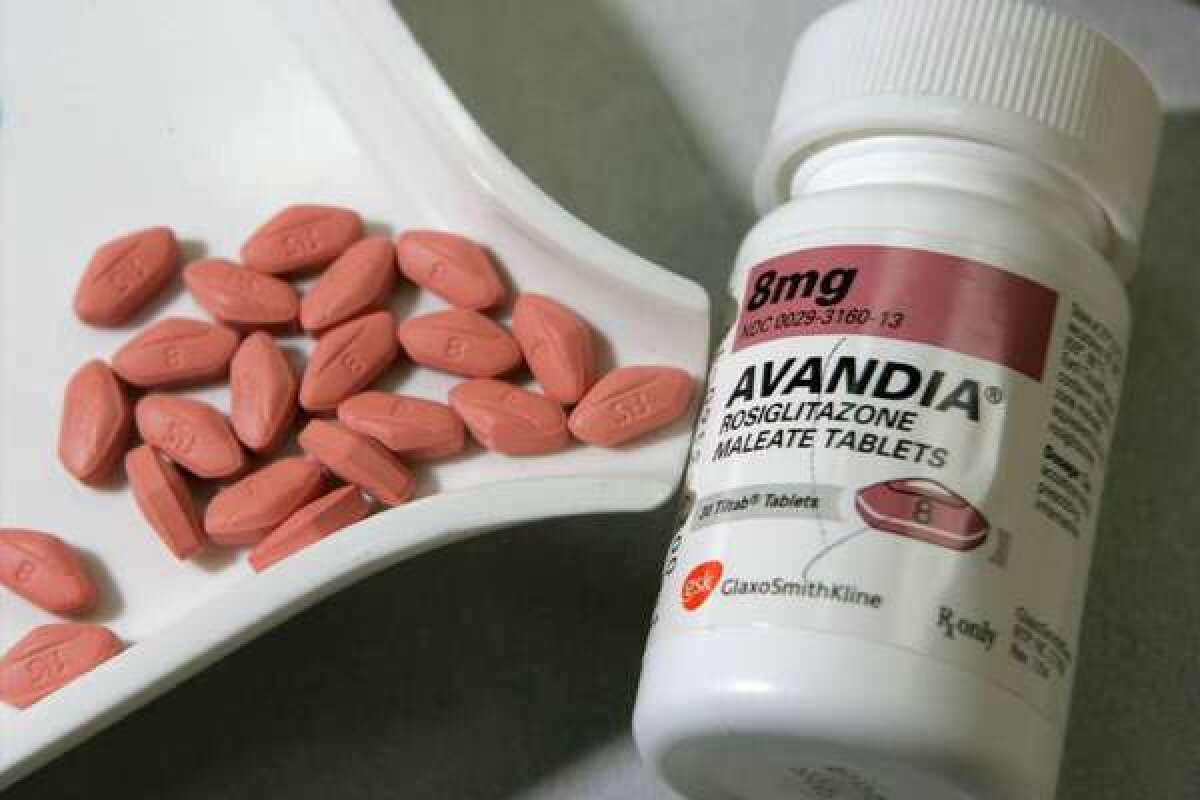
An advisory panel to the Food and Drug Administration on Thursday voted to keep the diabetes drug Avandia on the market with a slate of restrictions on its use, and suggested that although evidence of the drug’s safety problems may have been overblown, Avandia’s days as a blockbuster diabetes medication are probably behind it.
The vote came at the end of an extraordinary two-day meeting in which the FDA mounted a strong defense against skeptics who charged the agency had been hoodwinked about the safety of Avandia, also known by its generic name, rosiglitazone.
After the FDA ordered an independent reevaluation of a key clinical trial on Avandia’s safety, a majority of the panel’s members pronounced themselves reassured that the drug did not substantially increase heart attacks and deaths in those taking it. But several of the panel’s experts insisted that other studies of the drug’s safety had raised sufficient concerns that prescriptions should remain limited to patients with few alternatives and an understanding of Avandia’s potential risks.
Currently, 3,000 Americans with Type-2 diabetes take rosiglitazone, under strictures that require physicians who prescribe the drug and pharmacies that dispense it to be certified. Patients must also demonstrate that they have been notified of the drug’s potential risks and are taking it under close supervision. European regulators pulled the drug from the market in 2010, and a medication that was once the most prescribed treatment for Type-2 diabetes tumbled from its commercial perch.
Seven of the panel’s 26 voting members voted Thursday to recommend that the FDA lift all of those restrictions on prescribing Avandia.
Noting that FDA restrictions have denied many diabetics who might benefit from the option of taking Avandia, the panel’s lone patient representative voted to lift those strictures completely. “An overabundance of caution is not a neutral position,” said Rebecca Killian in explaining her vote Thursday. “Our charge here is to be safe, but sane.”
Only one panel member, Ida L. Spruill, voted to remove rosiglitazone from the U.S. market altogether. Spruill, a professor of nursing at Medical University of South Carolina, said she took Avandia for five years before being taken off the medication, and had lost confidence in the drug.
Of the 13 voting members who recommended changes to existing restrictions on those taking and prescribing Avandia, several said they did so in the hopes that Avandia’s maker, GlaxoSmithKline, would conduct new safety trials with the FDA’s blessing.
Tufts University cardiologist Marvin A. Konstam, who said he believed the FDA’s clamp-down on rosiglitazone prescriptions was appropriate in 2010, said he “would like to see a trial,” especially one that would measure Avandia’s safety and effectiveness head-to-head with that of pioglitazone, a medication in the same class as Avandia.
Conducting such a safety trial, given the existing restrictions on prescribing the drug, would probably hamper recruitment -- and would convey to many potential study participants that they were potentially signing up to take a drug with a clear record of heart attack risk, several panelists said. Others, however, expressed skepticism that a new study was in the cards.
“I think that train has left the station,” said Gerald van Belle, a University of Washington clinical trials specialist who voted to keep existing restrictions on rosiglitazone in place. Van Belle expressed doubt that a clinical trial of sufficient scale to dispel doubts about the drug “is feasible anymore.” With a welter of newer diabetes drugs in development or new to the market, “that’s where the action will be,” he added.
In addition, limits on GlaxoSmithKline’s rights to market Avandia without generic competition would make a major investment in new research on the medication unlikely. Since its introduction to the U.S. market in 2001, the British pharmaceutical firm has enjoyed market exclusivity, ensuring a substantial profit. But that is due to end soon.
The restrictions now in place on rosiglitazone’s use were imposed in 2010. They came after a team led by Cleveland Clinic cardiologist Dr. Steven Nissen published a culling of 42 small clinical trials and found that patients taking Avandia had a 43% higher risk than those on older diabetes medication. At the same time, U.S. government investigators turned up evidence that suggested GlaxoSmithKline, which sponsored a trial to assess rosiglitazone’s safety, biased the findings to discount or downplay cardiovascular events in those taking the drug.
That trial, called RECORD (short for Rosiglitazone Evaluated for Cardiovascular Outcomes and Regulation of Glycemia in Diabetes), got a thorough FDA-ordered review by a group of clinical trial experts at Duke University Medical School. The resulting “readjudication” was the centerpiece of the FDA advisory panel’s deliberations this week.
While discussion occasionally turned testy, the FDA panel’s two-day debate turned heavily on the fine methodological points of biomedical research. The panel’s researchers and clinicians debated at length the relative weight that physicians on the front lines of medicine should give to the findings of placebo-controlled trials and so-called meta-analyses, such as those conducted by Nissen. There was widespread agreement that the RECORD trial suffered from serious flaws in design. At the same time, the practice of gathering up small studies conducted separately and distilling their findings -- the design that pointed to safety concerns in Avandia -- also came in for harsh criticism.
Following Thursday’s vote, GlaxoSmithKline issued a statement thanking the panel and the FDA for its review. The company underlined that whether the evaluation team used standards in place at the time of the RECORD trial or new ones embraced by the FDA, the original findings of the company-sponsored research -- that heart attacks and deaths did not increase among those taking Avandia -- held.
“We continue to believe that Avandia is a safe and effective treatment option for Type-2 diabetes when used for the appropriate patient and in accordance with labeling,” said Dr. James Shannon, the company’s chief scientific officer.