At least three L.A. County patients infected after heart surgeries
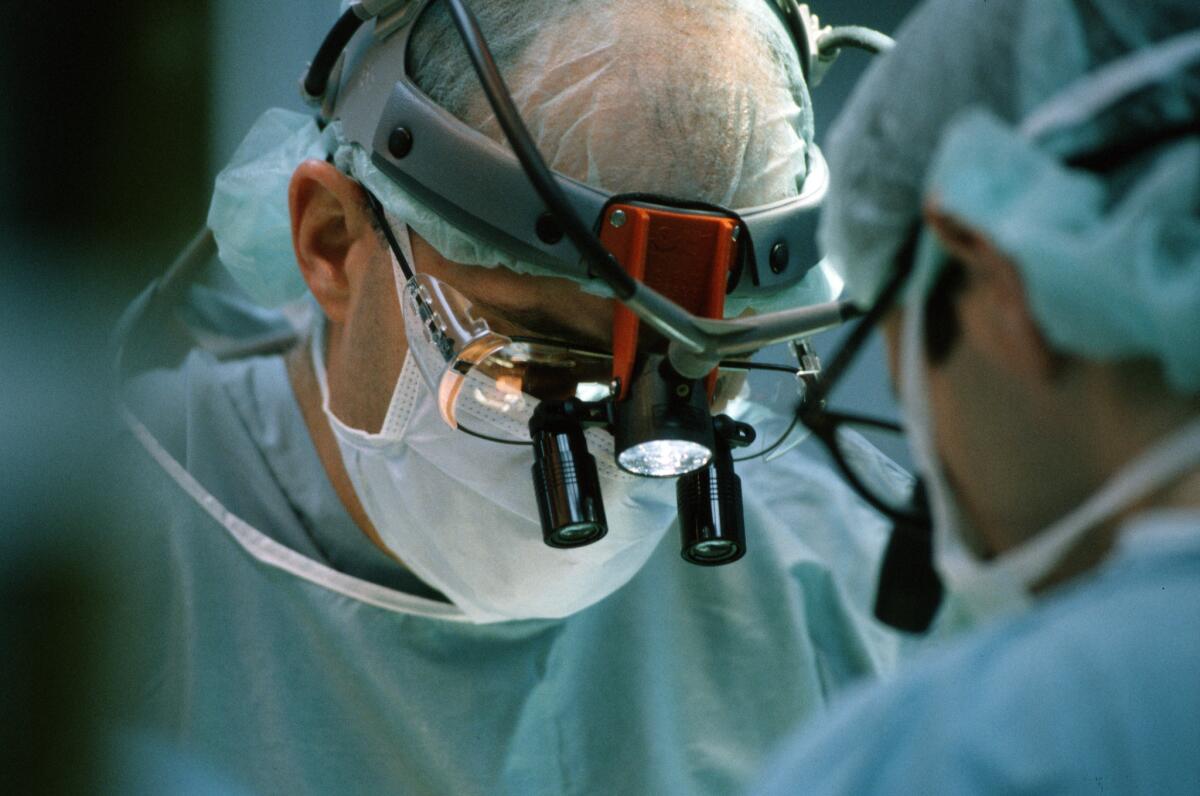
Los Angeles County health officials said Wednesday that at least three heart surgery patients at a hospital have been sickened by a dangerous bacteria linked to an operating room device.
Officials are now investigating a fourth patient infection at another hospital that may also be linked to the device, said Benjamin Schwartz, acting director of the county’s acute communicable disease control program.
All four patients had open chest cardiac surgery, he said, and survived their infections.
The device used in the heart bypass procedures has been linked to dozens of infections in patients across the country, including some who have died. Yet officials estimate that the risk of getting an infection is very low — between 1 in 100 and 1 in 1,000.
Schwartz said both hospitals were using a device that federal officials have said may have been contaminated with bacteria during manufacturing.
About 60% of heart bypass procedures in the U.S. use the Sorin Stockert 3T heater-cooler system, which helps keep a patient’s blood and organs at a specific temperature during the surgery, officials say.
Schwartz declined to name the two hospitals.
“It would be inaccurate to say these were the only hospitals where risks occurred,” he said.
Officials have talked to 24 hospitals in the county that use the device, he said. The two hospitals reporting the infections may have worked harder at trying to find patients who were sickened, he said.
Schwartz said the majority of the 24 hospitals told officials that they planned to notify patients who were at risk.
Infections caused by the slow-growing bacteria — Mycobacterium chimaera — are difficult to diagnose.
“The infections have occurred months or years following surgery,” Schwartz said.
Federal officials say patients who have had open heart surgery should seek medical care if they experience night sweats, muscle aches, weight loss, fatigue or unexplained fever.
The bacteria is common in soil and water, where it rarely makes people sick.
The heater-cooler devices have a tank containing water that is not intended to come into contact with the patient. But if the water is contaminated, the bacteria can become aerosolized in the operating room and enter a patient’s open chest cavity, the Food and Drug Administration has found.
The Sorin Group, which manufactured the device, changed its name to LivaNova in 2015 after a merger.
“We are working with regulators, clinicians and all relevant parties to resolve this important industry-wide issue,” Deanna Wilke, a LivaNova spokeswoman, said last month.
The FDA has found some infections tied to heater-coolers manufactured by other companies.
Schwartz said the county has asked all 24 hospitals using the Sorin heater-cooler to look for possible infections.
Some hospitals have now switched to heater-coolers manufactured by other companies, he said. Officials have advised those continuing to use the device to inform patients before their surgery that they are using a device linked to infections.
He said the heart surgery is so crucial for most patients that its benefits would outweigh the small risk of infection.
To read the article in Spanish, click here
Follow @melodypetersen on Twitter
More to Read
Inside the business of entertainment
The Wide Shot brings you news, analysis and insights on everything from streaming wars to production — and what it all means for the future.
You may occasionally receive promotional content from the Los Angeles Times.











