Hear me now? Gene therapy improves ‘bionic ear’ technology
A procedure that uses a series of electric jolts to inject lab-designed DNA molecules into cells of the inner ear may help to regrow auditory nerves in people with profound hearing loss, according to researchers.
In a paper published Wednesday in Science Translational Medicine, Australian researchers said they used tiny electrodes and gene therapy to regenerate nerve cells in chemically deafened guinea pigs.
The procedure, they said, may one day improve the functioning of human cochlear implants -- electronic devices that provide hearing sensations to the deaf.
“People with chochlear implants do well with understanding speech, but their perception of pitch can be poor, so they often miss out on the joy of music,” said senior author Gary Housley, a professor of neuroscience at the University of South Wales.
“Ultimately we hope that after further research, people who depend on cochlear implant devices will be able to enjoy a broader dynamic and tonal range of sound,” Housely said in a prepared statement.
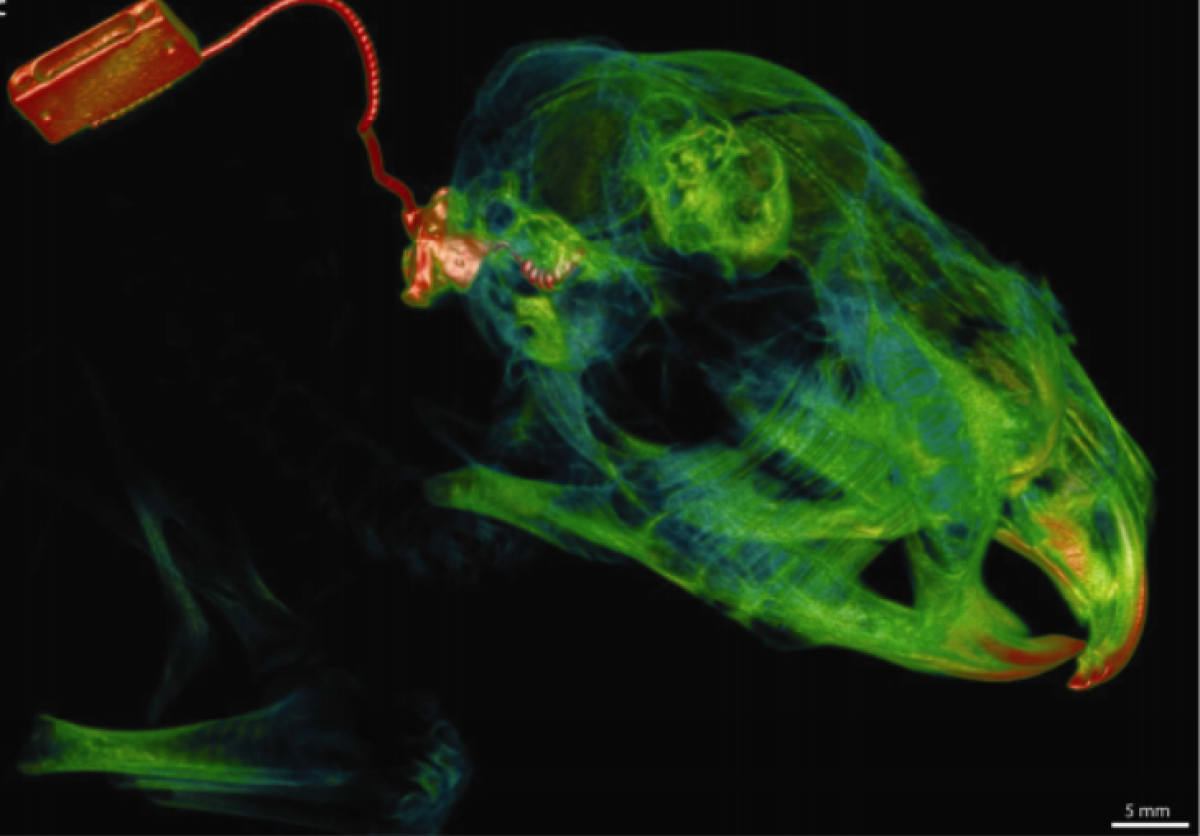
Houseley and his colleagues studied the procedure on guinea pigs because the structure of their inner ear is similar to that of humans.
The cochlea is shaped like a snail’s shell, and is filled with a multitude of tiny hair cells that move in response to sound vibrations. Those vibrations are then converted into electrical nerve impulses that are carried to the brain.
If the hair cells are lost or damaged due to age, genetics, chemical poisoning or loud noise, they will not grow back. In some people who are profoundly deaf, an electrode may be implanted within the cochlea that can stimulate some nerve cells.
While cochlear implants help roughly 300,000 patients throughout the world to detect and interpret speech, researchers believe they can be improved if nerve cells are encouraged to grow closer to the electrode. In this latest study, Housely and his colleagues set out to stimulate growth in spiral ganglion neurons in guinea pigs.
Study authors believed they could do this by causing inner ear cells to produce neurotrophins, proteins that control the development, maintenance and function of nerve cells.
Researchers injected short, ring-like sections of DNA, or plasmids, into the animals ears, in order to drive the expression of so-called brain-derived neurotrophic factor, as well as fluorescent marker genes. The scientists then used electrodes similar to cochlear implants to send between 4 and 100 volts of electricity into animal’s cochlea.
When cells are exposed to electric current, it causes their membranes to become extremely permeable, allowing the injected plasmids to enter the cell and direct production of the necessary proteins. This technique is known as close field electroporation, or CFE.
Study authors said that they observed regrowth of nerve cells in the ears of guinea pigs that underwent the electric pulse treatment, while those who did not showed no nerve growth.
When researchers tested the deafened guinea pigs two weeks later, they found that their cochlear implants were able to stimulate the new nerve cells and trigger a response in their brain.
While the study showed positive results in a short period of time, some questions remain about its effectiveness. Study authors observed a reduction in BDNF expression over three to six weeks.
Nevertheless, study authors said the electroporation techinque for gene therapy held promise for treating a variety of conditions.
“The development of electrode array-based CFE gene delivery may not only improve the hearing of cochlear implant recipients but also find broader therapeutic applications, such as in conjunction with deep brain stimulation, which uses electrode arrays similar to cochlear implant, to treat a range of neurological disorders, from Parkinson’s disease to psychiatric disorders.”