New coronavirus test is an imperfect step toward mass screening
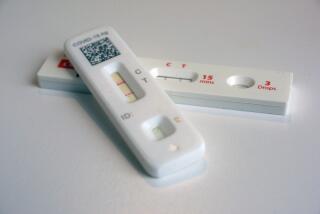
A new type of coronavirus test offers a cheaper, quicker way to screen for infections, moving the U.S. toward the kind of mass screening that experts say is essential to returning millions of Americans to school and work.
But the first so-called antigen test — approved Friday by the Food and Drug Administration under an emergency use authorization — is not quite the kind sought by top government health officials to identify patients with COVID-19. It is less accurate than the current gold standard for testing and can only be run on specialized equipment.
For the record:
1:01 p.m. May 12, 2020A previous version of this story said OraSure Technologies had a $700-million federal contract to develop a saliva-based coronavirus screening test. The contract is actually worth about $710,000.
“It is too early to tell” what kind of impact the new test will have, said Patricia Simner of Johns Hopkins University, who researches new ways of diagnosing infectious diseases. “It certainly has the potential to aid in more widespread testing.”
Here’s a closer look at the new test, which was developed by Quidel Corp. in San Diego.
What’s different about this test?
The test screens for new infections by looking for protein traces of the virus called antigens. Hospitals and doctors’ offices use the same approach for rapid tests for flu, strep throat and other infections. But they represent a trade-off, sacrificing some of the accuracy of more rigorous tests in exchange for quicker results at a lower cost.
“They allow you to dramatically expand testing, and they’re very cheap,” said Dr. Scott Gottlieb, the former FDA commissioner, in an interview Sunday with CBS. But he acknowledged that “they’re going to miss some patients who have COVID.”
Despite President Trump’s statements that the coronavirus was released from a laboratory in Wuhan, scientists say the evidence points to a natural origin.
Previously, the only way to diagnose active infections was a test that detects the genetic material of the virus. Although highly accurate, most of those tests take hours to develop on machines mainly found at commercial labs, hospitals or universities. Abbott Laboratories makes a 15-minute version of the genetic test that runs on its portable machine, but it only does one sample at a time.
Why is antigen testing important?
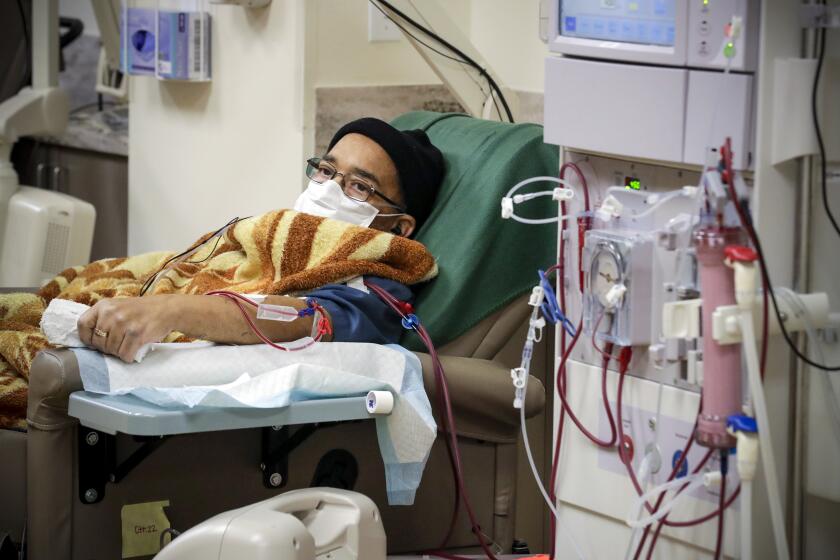
The U.S. is still struggling to increase testing to the levels that most public health experts say are essential. Harvard researchers have projected that the nation needs to be able to do 900,000 tests per day to be able to track new cases and contain new outbreaks as the country reopens. That’s more than three times the country’s current daily testing rate of about 275,000.
White House advisor Dr. Deborah Birx and other federal officials have said a “breakthrough” in antigen testing could open the way to daily testing before going to back to work or school.
The National Institutes of Health are pouring $1.5 billion into efforts to develop highly accurate, easy-to-use tests that could be performed without professional oversight or special equipment. Although the Quidel test is an important stepping-stone, it isn’t there yet.
How does it work?
The new test uses a nasal swab, like other screening tests, and delivers results in about 15 minutes.
The sample is put in a tube with testing chemicals and then into a cassette that goes into the company’s electronic reading device. There, it’s exposed to a testing strip embedded with laboratory-made antibodies, the specific blood proteins made by the body’s immune system when it detects an infection. If the antigens and antibodies interact, the test is positive.
How accurate is the test?
The new test is expected to detect about 80% of active COVID-19 infections, according to the FDA. That accuracy rate is similar to rapid antigen tests for seasonal flu.
“They are going to pick up fewer people that are infected,” Simner said. “So that’s where you see a lot of skepticism around using antigen tests for the diagnosis of COVID-19.″
For now, Simner and other experts said negative test results for people showing symptoms should be confirmed with the more accurate genetic test.
COVID-19 victims in their 20s and 30s make headlines, but does it mean the coronavirus has become more dangerous to younger people? Here’s what we know.
“But as you might imagine, that’s not a perfect scenario because you have to do twice as many tests when the result isn’t positive,” said Dr. Robin Patel, director of the Infectious Diseases Research Laboratory at the Mayo Clinic.
What’s next?
Many companies are working on tests that would be more accurate, easier to use and more suitable for mass screenings.
OraSure Technologies has a federal contract worth about $710,000 to develop a saliva-based antigen test that wouldn’t require specialized equipment or professional supervision. The company has similar tests for other viruses — including HIV and Ebola — and those have accuracy rates of over 95%. Along with accuracy, the company is focused on speed, aiming to delivers results in 20 to 30 minutes.
“If you are going to test people coming into work, you can’t make them line up at 3 a.m.” OraSure CEO Stephen Tang said. “You need to be able to get their results quickly.”