Column: The stem cell therapies offered by this La Jolla clinic aren’t FDA approved, may not work — and cost $15,000
- Share via
Jim Durgeloh, 59, was desperate to avoid surgery. After a career as a construction contractor and hours of leisure time spent on a motorcycle around his Longview, Wash., home, he was facing an operation to replace his left hip.
“That’s pretty invasive,” he said, nervous about a surgery that would require being cut open and implanted with an artificial hip; Jim’s brother had died from complications after a similar operation. In the search for an alternative, he and his wife, Janet, happened upon the website for La Jolla-based StemGenex Medical Group, which touts itself as “the world’s first and only Stem Cell Center of Excellence.”
But what caught the Durgelohs’ attention were the words of Rita Alexander, its chief administrative officer and a founder.
Alexander wrote that she had suffered debilitating rheumatoid arthritis until a stem cell treatment sent her into remission. “Today it remains my passion to advocate for those diagnosed with debilitating illnesses to have access to cutting edge stem cell treatment,” she wrote.
“Rita was very inspiring,” Janet Durgeloh says.
We have anecdotal feedback based upon direct reports from our patients that their symptoms have improved and their quality of life has increased.
— Jamie Schubert, StemGenex spokeswoman
Durgeloh’s doctor in Washington was skeptical about the therapy offered by StemGenex. “He didn’t think it was going to work,” Durgeloh says. The therapy isn’t approved by the Food and Drug Administration, which says such treatments are “not based on scientific evidence” and can be unsafe. Then there was the cost: about $15,000, not including airfare. That wasn’t covered by Durgeloh’s insurance, which would have paid for his hip replacement.
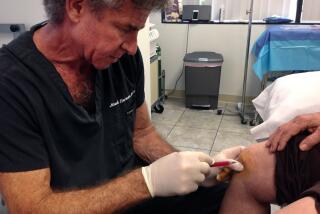
But on a recent Wednesday morning, the Durgelohs were at the DoubleTree hotel in Del Mar, where their bill was paid by StemGenex. Durgeloh was still wearing a bandage on his midriff, where one day earlier a StemGenex doctor had performed liposuction to obtain stem cells that subsequently were reinjected into his body, ostensibly to regenerate his damaged bones and tissues. They were preparing to fly home, infused with the hope communicated by the clinic staff, who “seemed very optimistic,” Durgeloh told me.
A lawsuit in San Diego federal court suggests that StemGenex may have given the Durgelohs nothing but hope. Three StemGenex patients — two with diabetes and one with lupus — say they were misled by the medical group’s marketing pitch to pay $14,900 each in 2015 and 2016 for therapies that have had no effect.
The lawsuit, which seeks class-action status, claims that StemGenex has made its money by “targeting the ill and the elderly” with “false, fabricated and purposefully misleading” claims about patient satisfaction. Selena Moorer, a lupus patient from Florida, and her two co-plaintiffs say StemGenex has “no reasonable basis for its marketing claim that the Stem Cell Treatments were effective to treat diseases as advertised.” The lawsuit names StemGenex, Alexander and Andre Lallande, the group’s chief medical officer, as defendants. The company denies the claims made in the lawsuit.
Durgeloh’s treatment was typical of the procedures offered as stem cell therapy. He says he received injections directly into his hips, his ailing knees and his back, with whatever was left over suffused into his body via an IV drip.
What’s most important to know is that there’s no accepted scientific evidence that treatments using cells from adipose fat tissue layers work.
But as we reported last year, many clinics offering the treatments capitalize on the public’s impression that stem cells have become some sort of medical miracle. Dr. Mehmet Oz warned his vast television audience about this misconception in February, when he aired a lengthy undercover investigation of stem cell clinics and called for government regulation. StemGenex wasn’t mentioned in the piece.
StemGenex, in its reply to the Moorer lawsuit, asserts that the plaintiffs “cannot prove” that its “representations regarding the efficacy of its stem cell treatments are actually false.” The plaintiffs, it continues, “do not cite to a single scientific study that disproves [StemGenex’s] advertised claims.”
StemGenex may not have to prove that in a court of law, but that’s not the way federal regulation works. At nearly $15,000 a pop, the companies should have to show a treatment works.
The FDA has been grappling with this very point in pondering how to regulate the burgeoning industry. There are more than 500 clinics offering stem cell treatments in the U.S., according to a survey released last year by stem cell scientist Paul Knoepfler of UC Davis and bioethicist Leigh Turner of the University of Minnesota.
Right now, there’s no consensus how these clinics should be regulated.
In 2015, UC San Diego researchers described stem cell treatment as “medicine’s Wild West.” As Hermes Taylor-Weiner and Joshua Graff Zivin observed, “Because FDA guidelines are ambiguous, stem-cell clinics have in effect been operating without regulation.”
The proliferation of the clinics has forced the FDA to take a closer look.
The government agency maintains that using stem cells extracted from a patient’s fat requires licensing as a drug, device or biological product, which means the clinics have to demonstrate the products are safe and effective, possibly via a clinical trial.
The clinics obviously disagree. Steven Brody, chief scientific officer of StemGenex, testified at an FDA hearing in September that if the FDA took a hands-off approach, “this would help our patients have access to stem cell therapies.”
Earlier this month, the New England Journal of Medicine reported the “devastating outcome” for three elderly women injected with fat-derived stem cells directly into their eyeballs by a clinic in Florida as a treatment for macular degeneration. The treatment left the patients totally or mostly blind.
Stem cell clinics typically are cagey about what patients should expect. They “neither claim their treatments are effective nor explicitly state that they’re unfounded,” Taylor-Weiner and Zivin observed. “Their language is intentionally imprecise and exploits the vulnerability of patients with debilitating diseases.”
Indeed, a disclaimer on the StemGenex home page states, “Stem cell therapy is not FDA approved,” and, “StemGenex Medical Group and affiliates do not claim that treatment using autologous stem cells are a cure for any condition, disease, or injury.”
That’s a striking admission for a treatment costing nearly $15,000 out-of-pocket and might help explain why health insurers shun the treatments.
The emotional video testimonials from patients posted on the StemGenex website carry disclaimers that the results experienced by those patients “may not be typical or expected. … You should not expect to experience these results.”
When I asked Jamie Schubert, a StemGenex spokeswoman, to point me to a scientific study or any other evidence that its treatments work, she replied that “anecdotal feedback” from patients indicates that “their symptoms have improved and their quality of life has increased.”
There are other red flags. One of the medical group’s physicians, plastic surgeon Scott Sessions, was placed on three years’ probation by the California Medical Board in February. He was accused of negligence related to cosmetic surgery and other procedures he performed on two patients at an unrelated facility in 2011 and 2013.
Schubert told me Wednesday that “Dr. Sessions has informed us that he is in compliance with all requirements of the probationary terms of the medical board.” But the very next day, his name, photograph and bio had disappeared from the StemGenex website. Sessions didn’t respond to a request for comment.
The same thing happened with the logo of the American Board of Surgery, which had been prominently displayed on the StemGenex site, implying the company had the certification board’s seal of approval. After I mentioned to Schubert that a board official told me that display was “a complete misuse of our logo,” it vanished. Schubert called it “an error.”
People’s health needs are not suitable for unregulated Wild West experimentation, and anecdotal feedback isn’t proof that “cutting edge” treatments are safe and effective. The course couldn’t be clearer for the FDA and state medical regulators across the country: If these stem cell clinics are endangering their customers’ health and draining their pocketbooks for quack remedies, shut them down.
Keep up to date with Michael Hiltzik. Follow @hiltzikm on Twitter, see his Facebook page, or email michael.hiltzik@latimes.com.
Return to Michael Hiltzik’s blog.