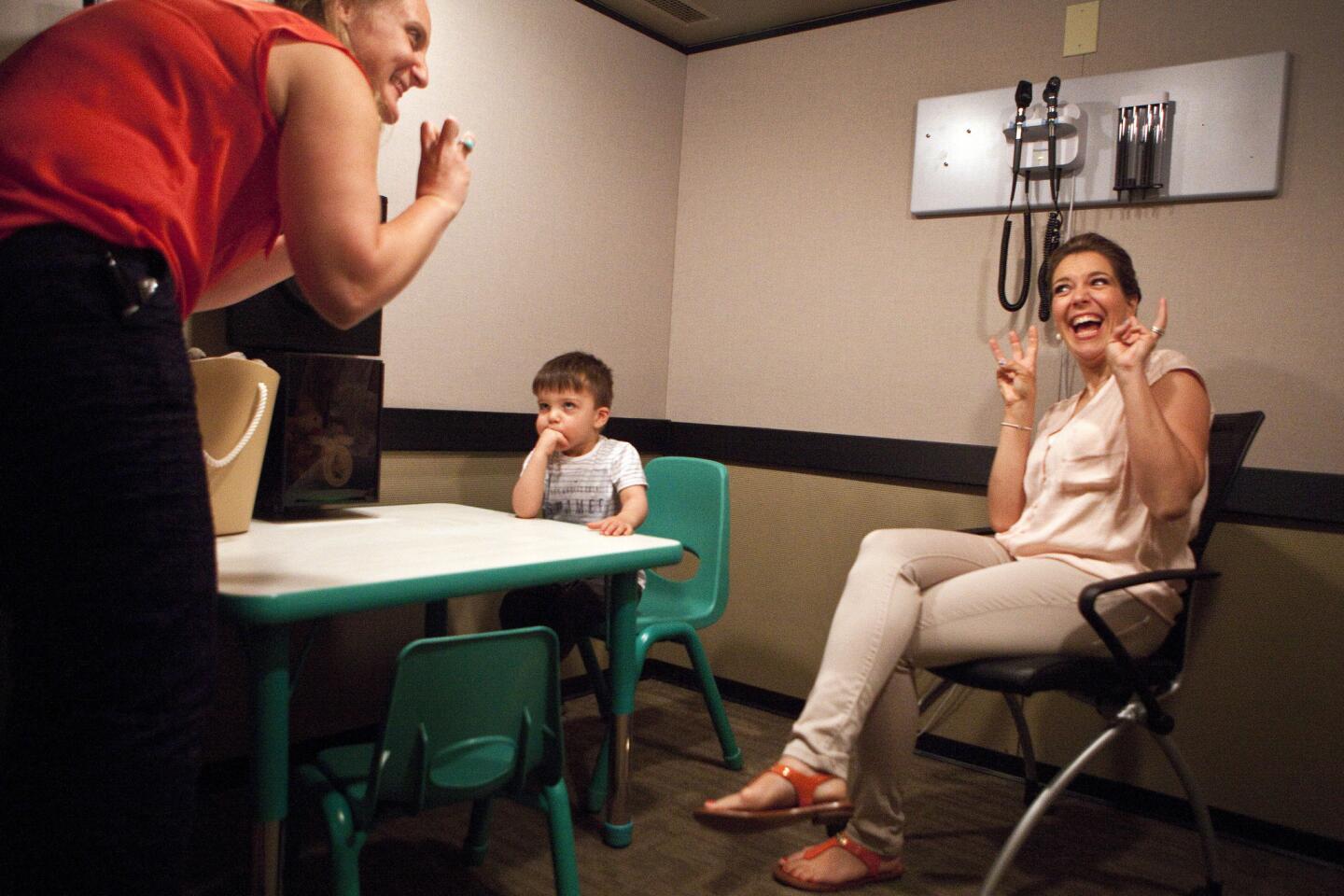Great Read: For a 3-year-old boy, a risky operation may mean a chance to hear
- Share via
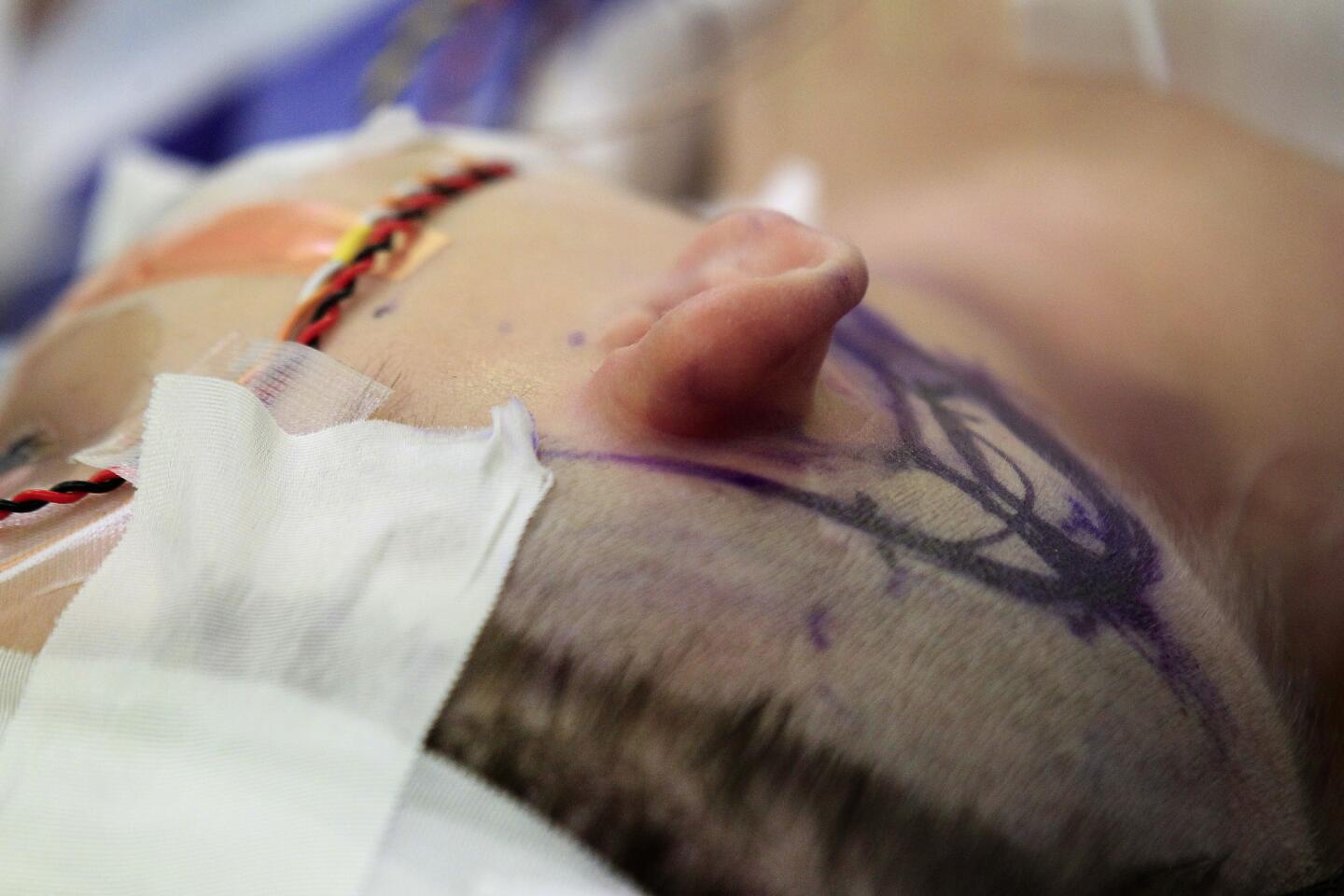
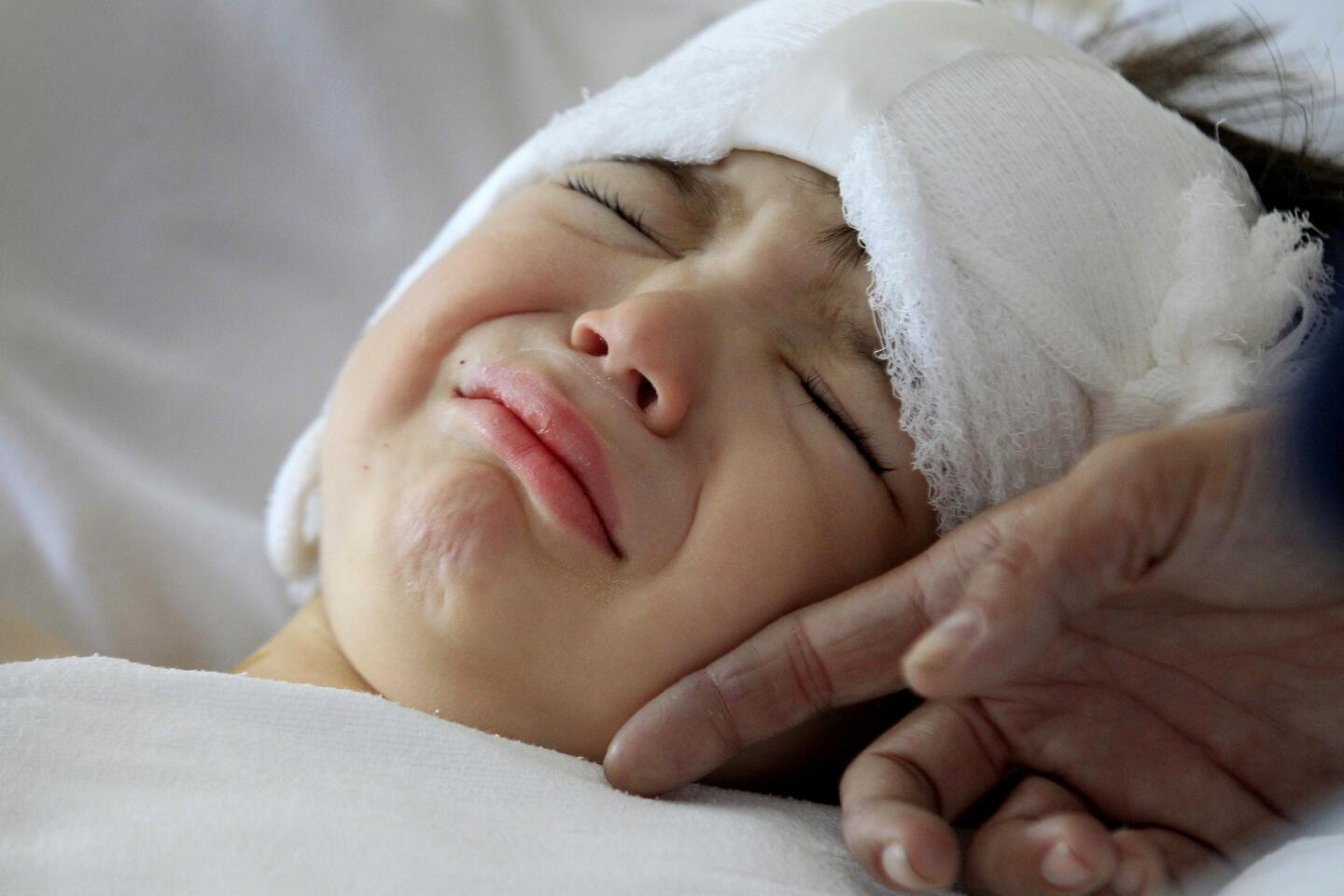
Auguste is 3 years old, a charmer with big blue eyes, long lashes and a playful smile. He’s wearing a T-shirt that says “Make some noise” and fiddling with his Etch A Sketch in a hospital exam room.
But when a doctor reaches toward his temple, he quickly releases the knobs and tilts his head. He knows what they’re looking for. Behind each ear, under the skin, are cochlear implants — surgically implanted devices meant to help process sound.
But they didn’t help Auguste.
He was born without an auditory nerve, the wire that ferries sound from the ear to the brain. He points out planes in the sky but can’t hear their engines roar. He’s never heard the voices of his 4-year-old sister or 1-year-old brother. He doesn’t know the sound of his own name.
Now, a team of Los Angeles doctors and researchers believes it can help Auguste hear. But the device the team is testing will require surgeons to go much deeper into Auguste’s skull, all the way to the brain.
Sophie Gareau, Auguste’s mom, often tears up when she thinks about what’s ahead.
“He’s so perfect, you know?” she says. Her voice breaks. “I don’t want anything to happen to him. But I am so convinced that it’s going to work.”
::
Sophie realized something was off when Auguste was 6 months old: Her baby wasn’t babbling. Doctors in Montreal said he was fine. She insisted otherwise, but it was nearly a year later before audiologists played sounds as loud as an airplane engine in the toddler’s ear and confirmed what his French Canadian parents had suspected: Auguste was profoundly deaf.
“I think it takes a moment to sink in and then you cry for a week,” Sophie said.
Audiologists put him on track for the cochlear implant, which places electrodes in the snail-shaped cochlea, part of the inner ear, to stimulate the auditory nerve. It’s a common procedure for deaf children: About 38,000 children in the U.S. have received them.
But the surgery didn’t work. Auguste had a cochlea, doctors discovered, but no auditory nerve to deliver sound to the brainstem.
Sophie and her husband, Christophe Majkowski, were crushed, but they focused on the positive. “He’s perfectly healthy, and he’s smart and he’s quick,” said Sophie, who shares her son’s big blue eyes and constantly chats with him in sign language.
She’d heard about something called an auditory brainstem implant, or ABI. The device uses an external processor on the ear to send sound to a surgically implanted electrode array. A doctor warned her against it.
“He had mentioned that only the Italians were doing it and they were ‘cowboys,’ which in French means ‘crazy people,’” she said.
The Italian in question was Vittorio Colletti, who began putting the devices in healthy deaf patients in the late 1990s at the University of Verona. This was unheard of at the time: There had to be an urgent reason — such as removal of a dangerous tumor — to perform such a risky surgery.
Colletti was criticized. But he began reporting incredible results; some patients with the device could even conduct business on the phone, without lip-reading.
The medical community was skeptical. So Colletti invited Robert Shannon, who helped develop the modern brainstem implant while working as a medical researcher at the House Research Institute in Los Angeles, to see for himself.
Shannon spent hundreds of hours testing patients in Italy.
“I was astounded,” said Shannon, who now works at USC. “It was even better than he said.”
A team of researchers that included Shannon wanted to introduce the procedure to children in the U.S. But a risk-averse Food and Drug Administration denied the team’s initial request, said USC audiologist Laurie Eisenberg, one of the lead scientists.
“After all,” Eisenberg said, “it is brain surgery in children.”
After some key changes in FDA personnel, the team received the go-ahead in 2012. In spite of several setbacks, the team pressed on, because the National Institutes of Health had agreed to pay for the team’s clinical trial.
Auguste is the first patient.
::
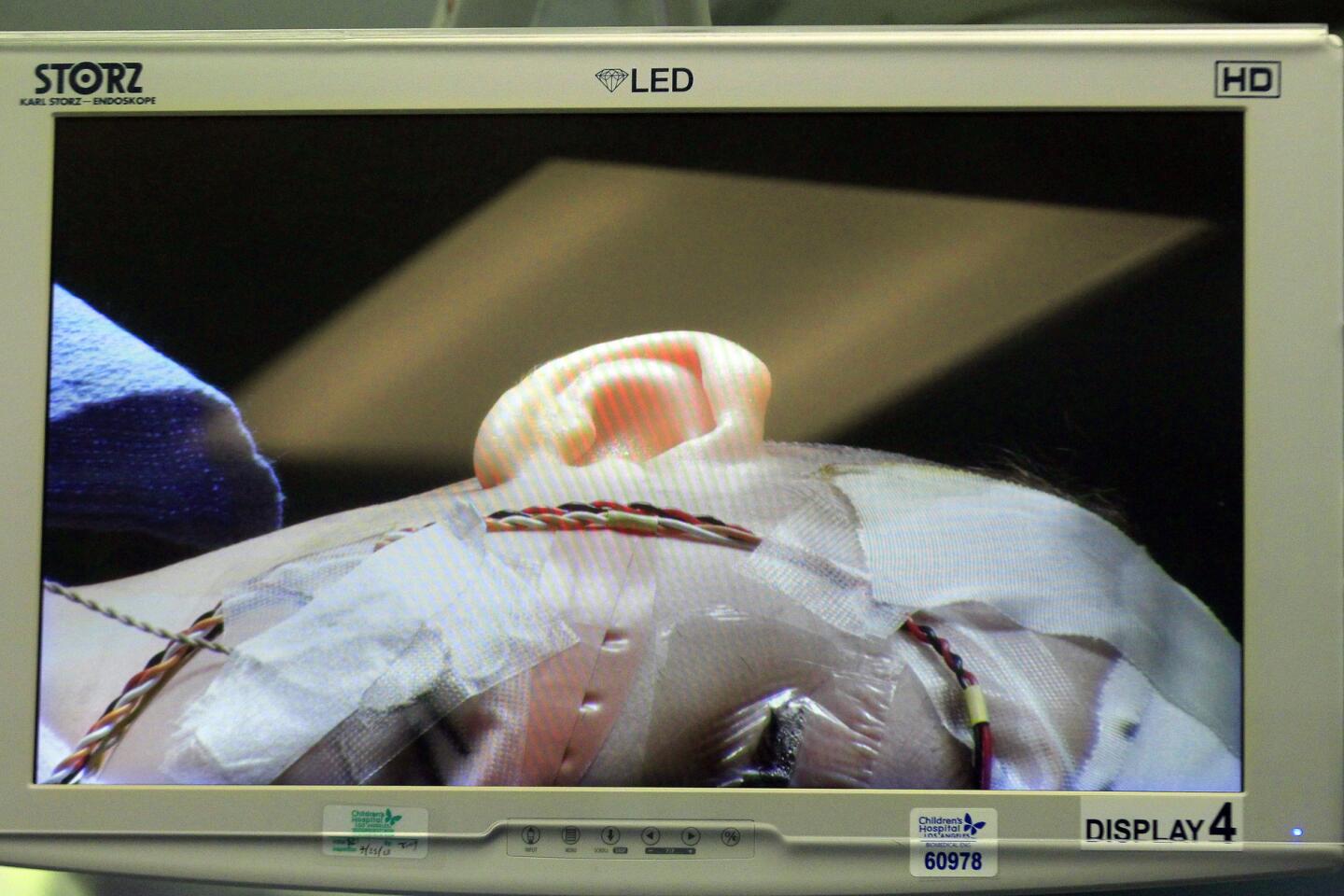
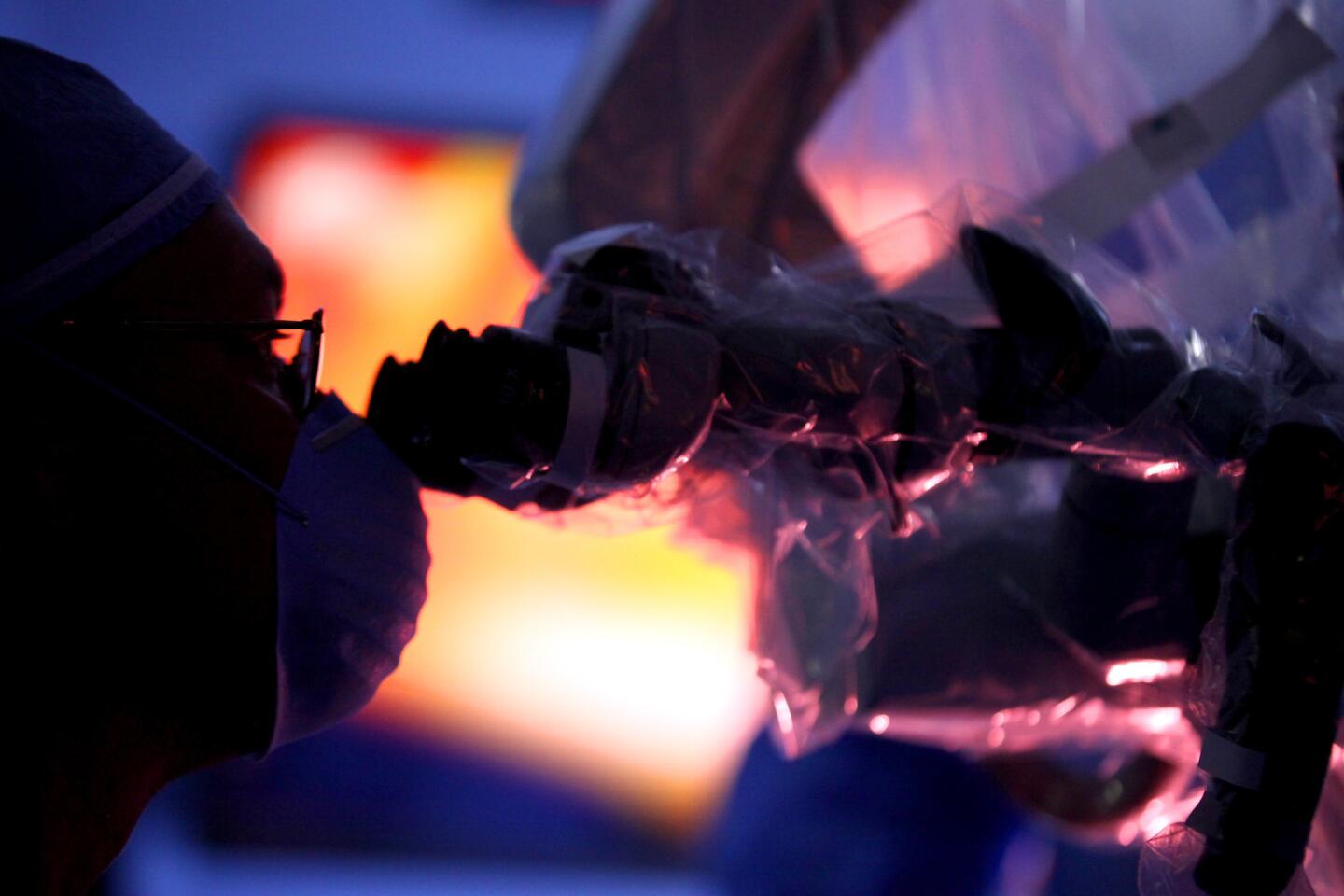
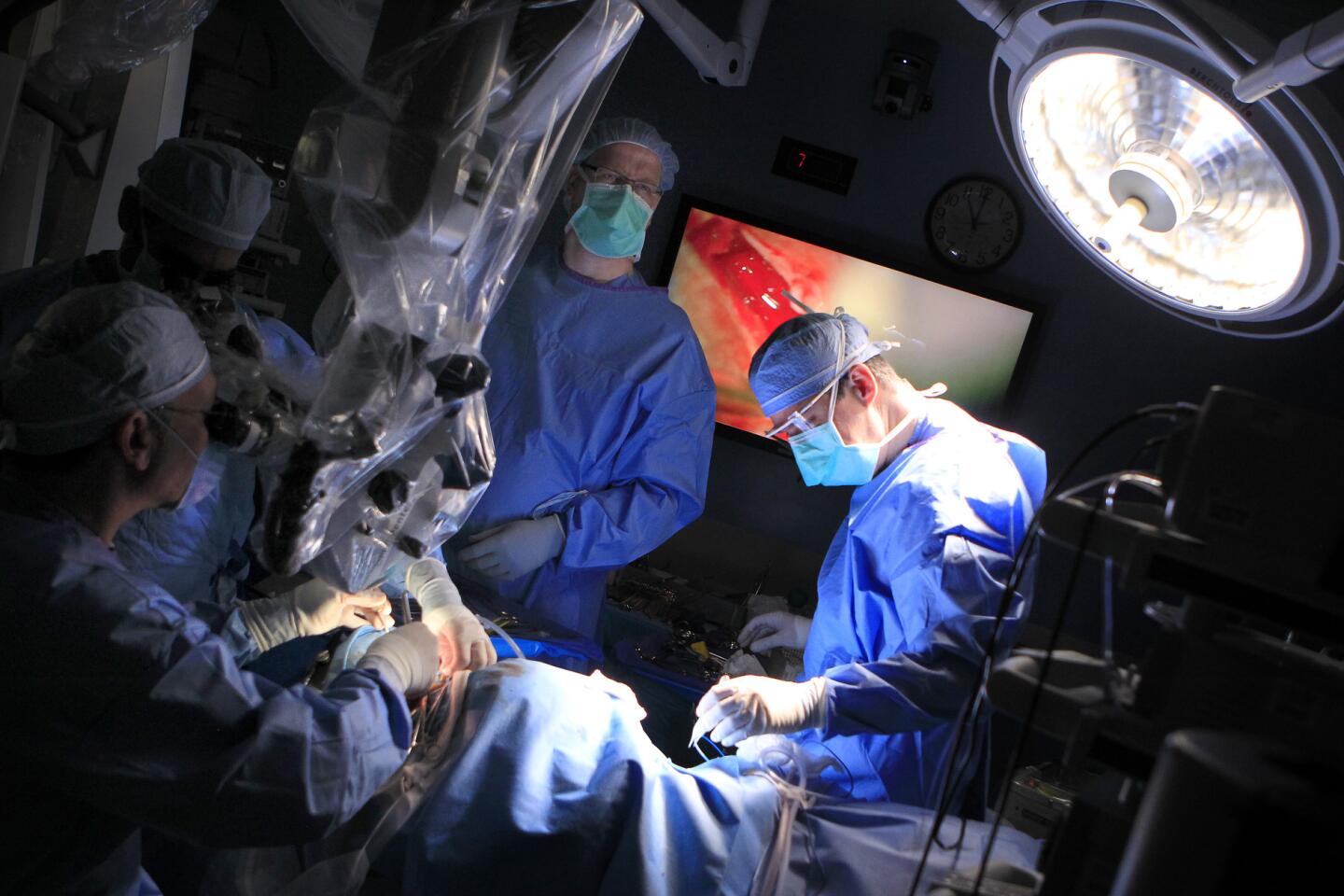
Auguste lies unconscious in the operating room at Children’s Hospital Los Angeles, a blue sheet covering his body. The only part of him visible is a small square where the three surgeons are operating — their window into Auguste’s brain.
Pediatric neurosurgeon Mark Krieger stands over this tiny red window, gloved hands at work. Marc Schwartz, a neurosurgeon with the Los Angeles-based House Clinic, speaks in a voice muffled by the mask they all must wear. His colleague Eric Wilkinson, one of the lead scientists, trains a plastic-wrapped microscope on the exposed flesh.
The surgeons cut through skin, saw through bone and snip through the dura, the brain’s protective covering. They drain cerebrospinal fluid so that the cerebellum relaxes, allowing them to move past the bulging tissue and reach the brainstem. Occasionally a spurt of blood runs across the opening, but the surgeons don’t flinch.
Colletti, the “cowboy” who pioneered the procedure, is sitting nearby. He has flown in from Italy to see the clinical trial begin. He can’t wield a knife; all he can do is offer advice as he watches Auguste’s brain on a large screen.
Schwartz knows the path to the cochlear nucleus, the spot on the brainstem where the ABI will go. He’s seen Colletti perform the procedure in Italy about half a dozen times. He follows the ninth cranial nerve to the boundary between the cerebellum and brainstem, then looks for a tiny nook called the lateral recess. That’s where the cochlear nucleus should be.
But it isn’t.
“There’s nothing there. There’s no hole there,” Schwartz says. “No hole. Not even anything that looks like a hole. Remotely.”
The chatter in the room has dropped to hollow silence, punctuated by the harsh beep of the monitors.
Schwartz retraces his steps, over and over. No luck. The surgeons debate what to do next. If they can’t find the lateral recess, they can’t put the device in.
Wilkinson urges him forward, but Schwartz is worried.
“I’m just afraid of hurting the kid,” he says.
At last, Schwartz finds the entrance. Over several minutes, he carefully inserts the 8-millimeter-long electrode array into the right spot.
“Bravo,” Colletti says. “Bravo, bravo!”
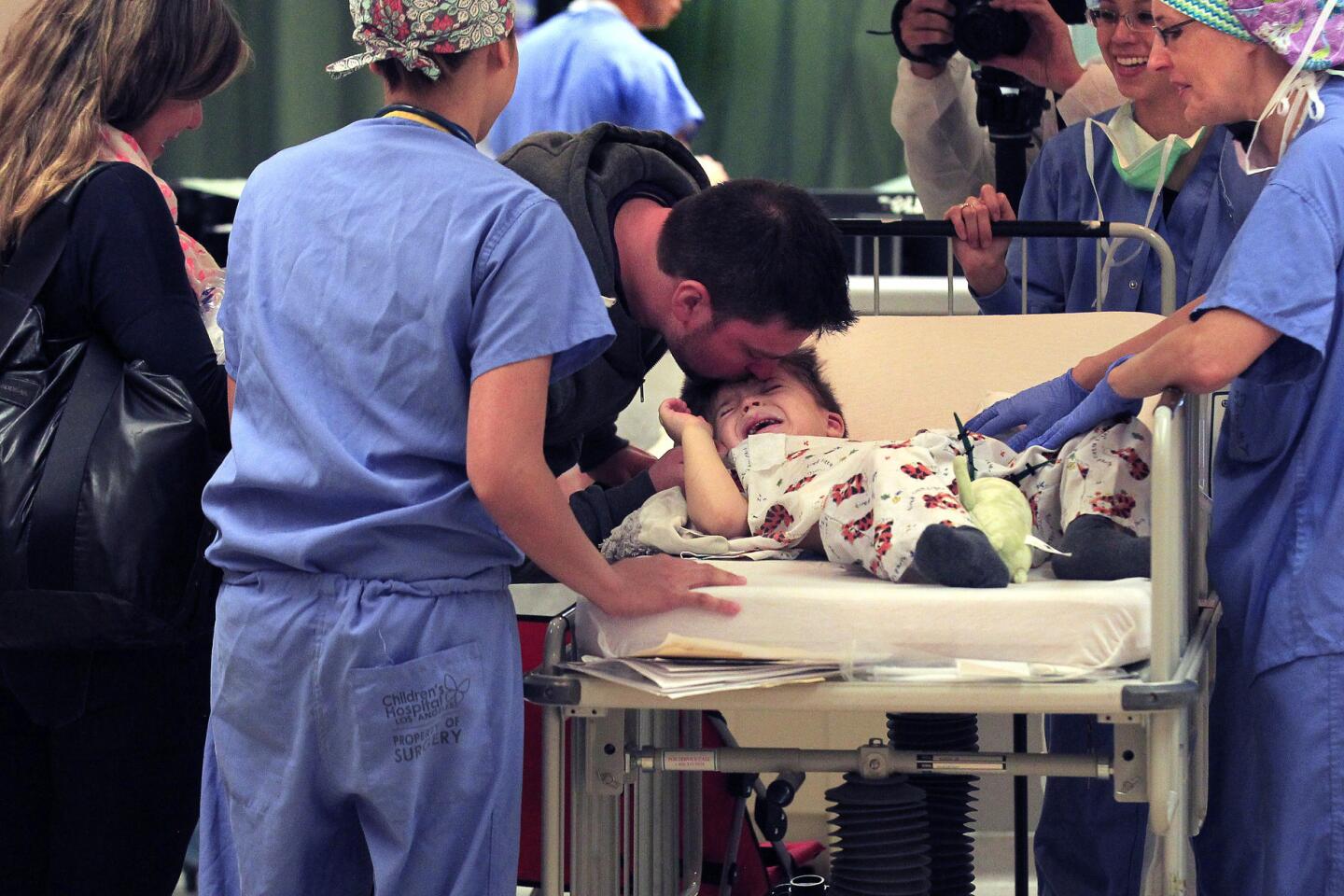
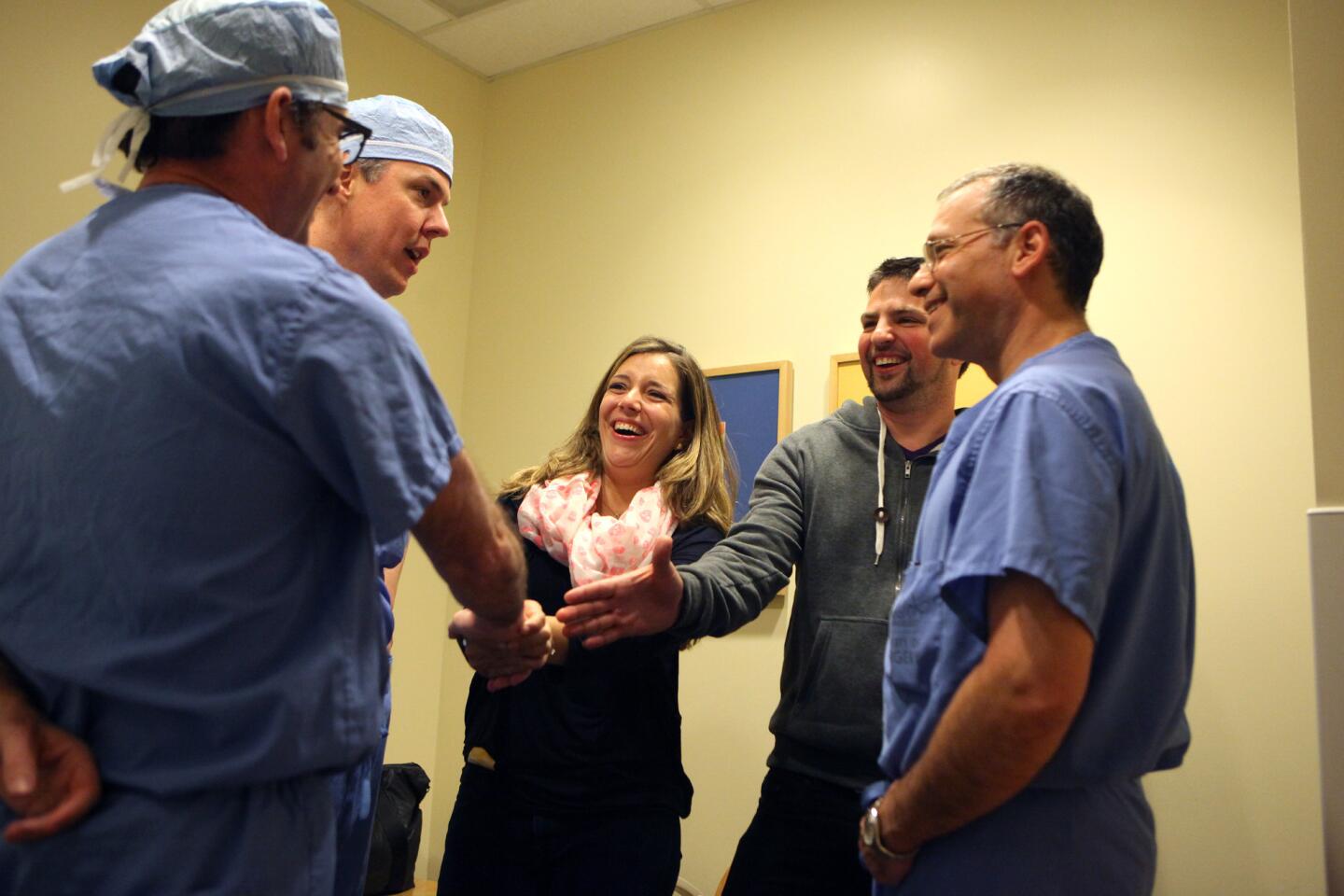
After the roughly six-hour operation, the doctors find Auguste’s parents in a waiting room. The surgery went well, they say. Auguste will be awake soon.
Christophe and Sophie hug each other tightly. When the surgeons are gone, she lets out a whoop, edged with exhaustion.
::
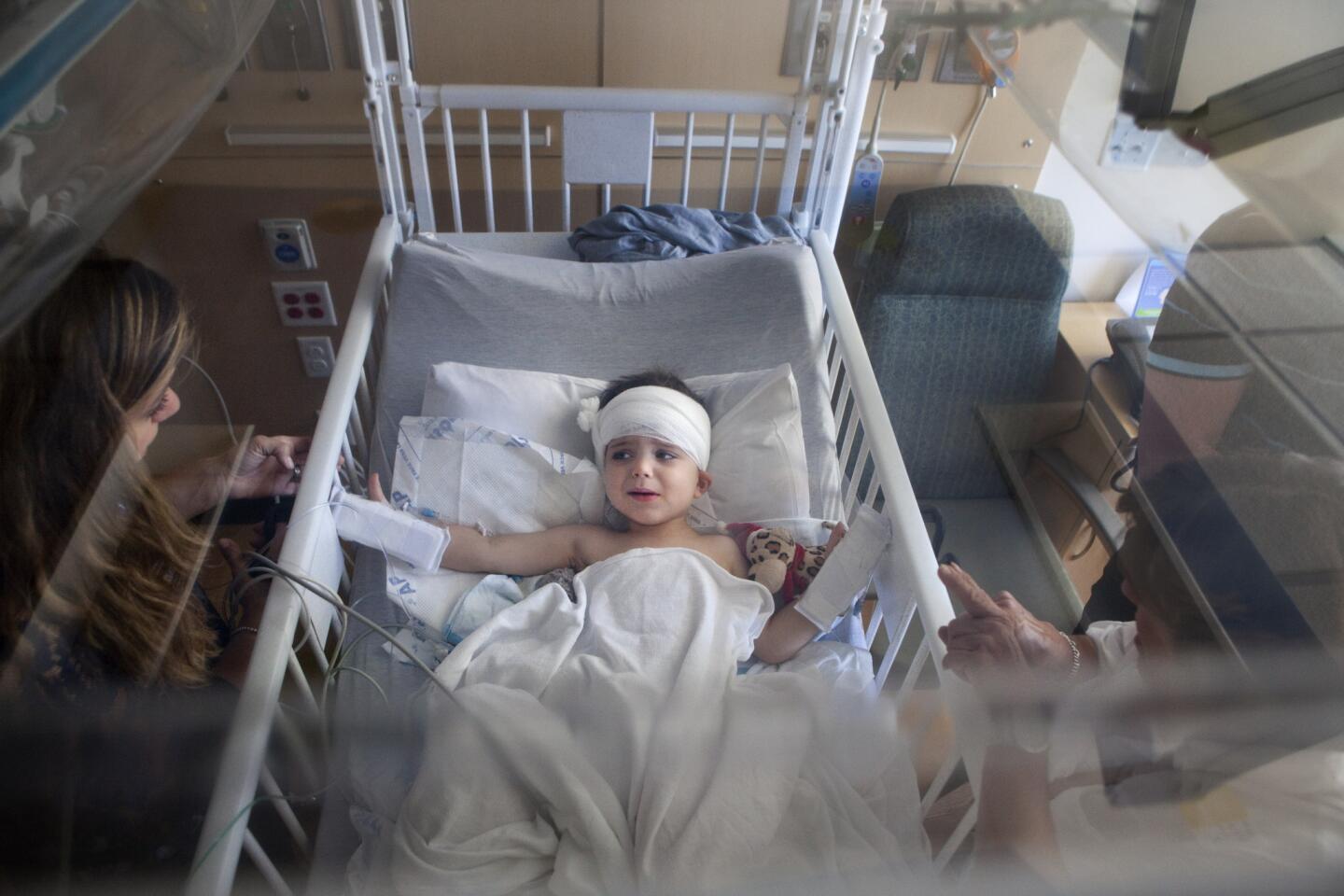
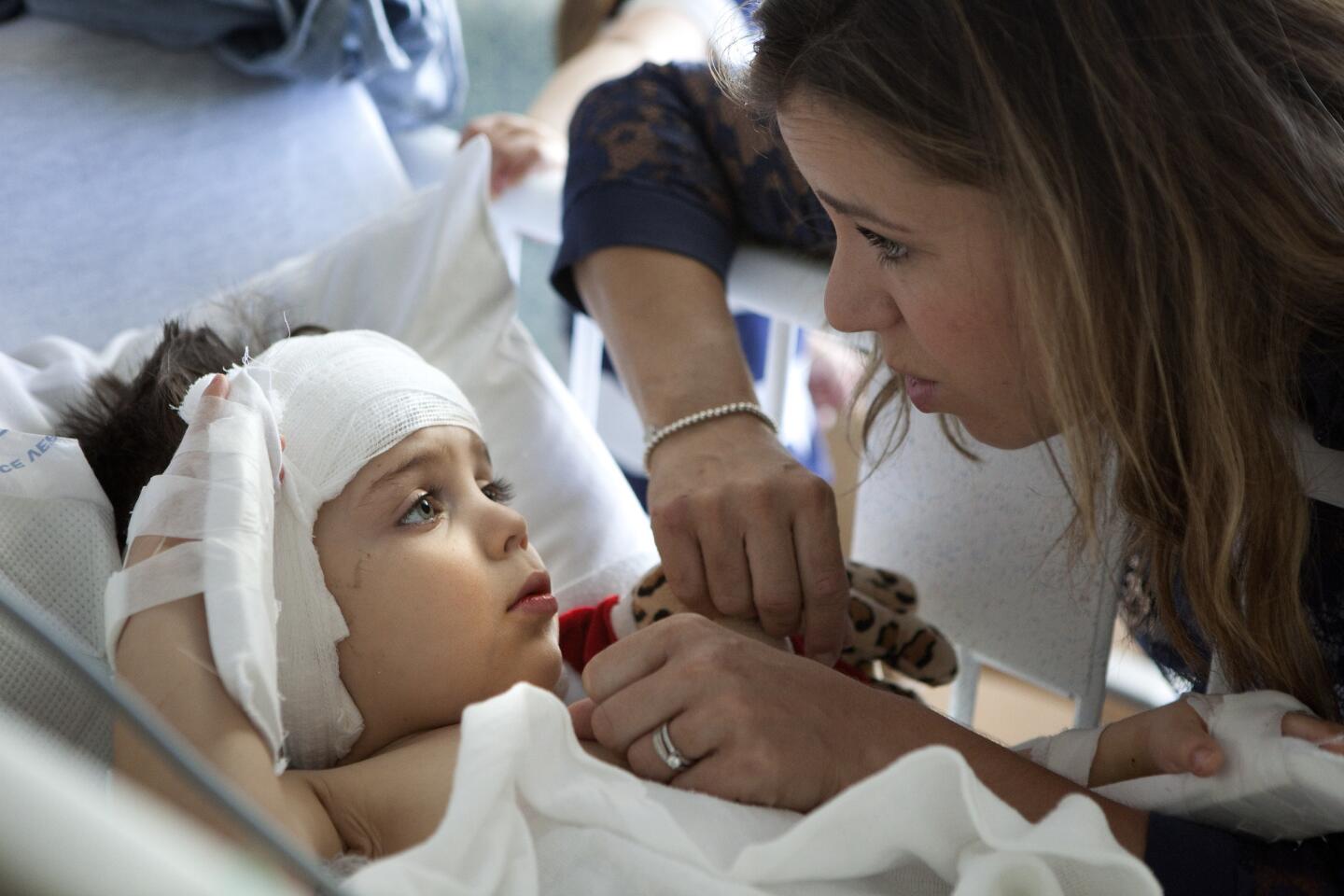
There are a few scary days for Auguste’s parents. Soon after the surgery, Sophie discovers blood seeping onto his pillow.
A week passes before Auguste can walk again. A few weeks later, he’s playing in the sand at Venice Beach with his family, a new haircut outlining the pink crescent scar behind his ear.
Inside Auguste’s head, his brain is healing and fibrous tissue is growing around the device. Outside, he’s flailing at his older sister, Daria, as they both try to climb onto their dad’s back. Christophe rears like a horse and they slip into the sand, giggling and out of breath.
Sophie says she’s keeping her expectations in check. The ABI delivers more rudimentary sound than does natural hearing. Auguste will always be deaf, and they’ll always sign with him, she says. If he never speaks or experiences the nuances of music, that’s fine.
All Auguste needs are basic reference sounds — the honk of a horn in a busy street, the clatter of something falling behind him — to connect him to the world and keep him out of harm’s way.
::
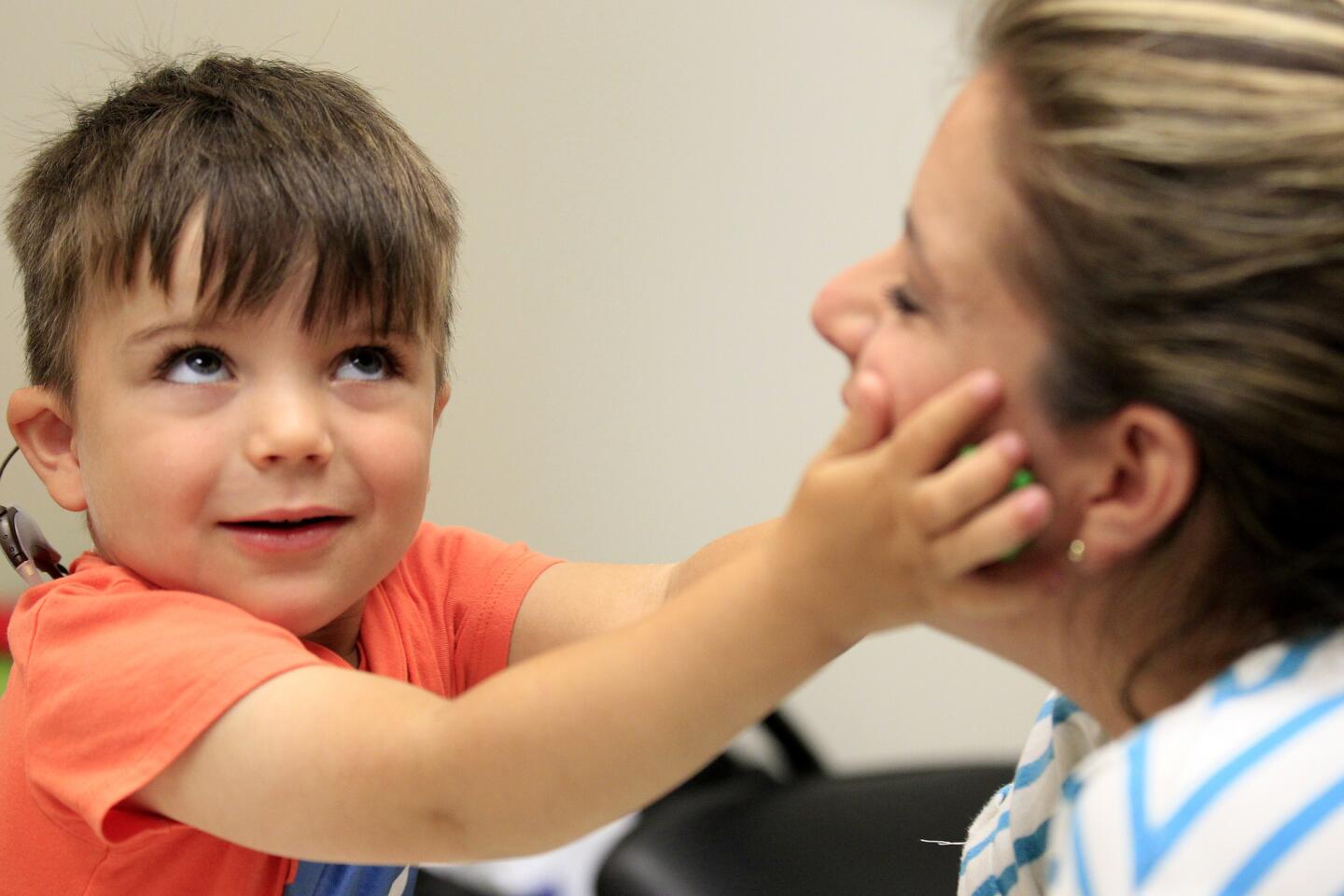
Auguste sits at a toy-filled table at USC’s Keck School of Medicine. After a month of recovery, it’s time to find out whether the surgery worked.
Margaret Winter and Jamie Glater scrunch around the kid-sized table. They’re known as “child whisperers”; because deaf children can’t say exactly what they’re hearing, the USC audiologists have to be able to read their reactions, such as a quick glance or a sudden stillness.
As Auguste plays with his trucks, Winter uses her computer to remotely activate individual electrodes on the device, each of which represents a different pitch range. If the device works, Auguste should hear these simple beeps.
Auguste is rolling a toy monster truck across the table. As the beep sounds, he freezes, mid-car crash. Something’s happened.
Glater makes a crooked L with her fingers, the thumb pointed at her ear. “Listening! You’re listening!” she says. Auguste searches their faces, then goes back to his toys.
Delighted, the audiologists go through several more electrodes, dialing up the electrical signal on each until they see a clear reaction. Then, it’s time to try a human voice. They turn on the microphone.
“Bap-bap-bap-bap,” Glater says. Every time she says the strong syllables, Auguste drops a token in a Connect Four grid, as he’s been trained to do. The adults cheer.
Winter turns the signal up. Auguste winces — the way you do when a sound is too piercing— and brings his hand to his head, then runs into Sophie’s open arms. He shakes his head so hard, the earpiece flies off.
That’s perfectly normal, Winter tells the parents. It’s a completely new experience — more of a sensation than a sound at first. Whatever he felt, it “penetrated his world,” Winter says.
Over time, the brain picks up more complex information, teases apart different pitches, learns to tell long, smooth sounds apart from short, staccato ones. How it manages this incredible feat is a mystery that researchers are still trying to solve.
Making that progress will take years of therapy and training for Auguste to learn how to process sound and, if possible, to speak.
But first, baby steps. The team leaves the device on at a low level. The next day, they try again.
“Hey, Auguste,” Sophie says. At first, he whines, but then he reaches for another toy. He’s getting used to it.
Glater grabs a toy train. “Choo choo choo,” she says. Christophe chimes in. “Choo choo choo,” they chorus. Auguste giggles. “You like that!” Sophie says.
After the testing, Auguste plays in the hallway. And when the device falls off his ear, he stops in his tracks. He picks it back up. He wants it on.
Twitter: @aminawrite