FDA approves new opioid painkiller said to last 24 hours
- Share via
Federal regulators gave the green light to a once-a-day opioid pain-relief pill that is up to 24 times more powerful than a single Vicodin but is designed to thwart the immediate release of its full — and potentially lethal — payload.
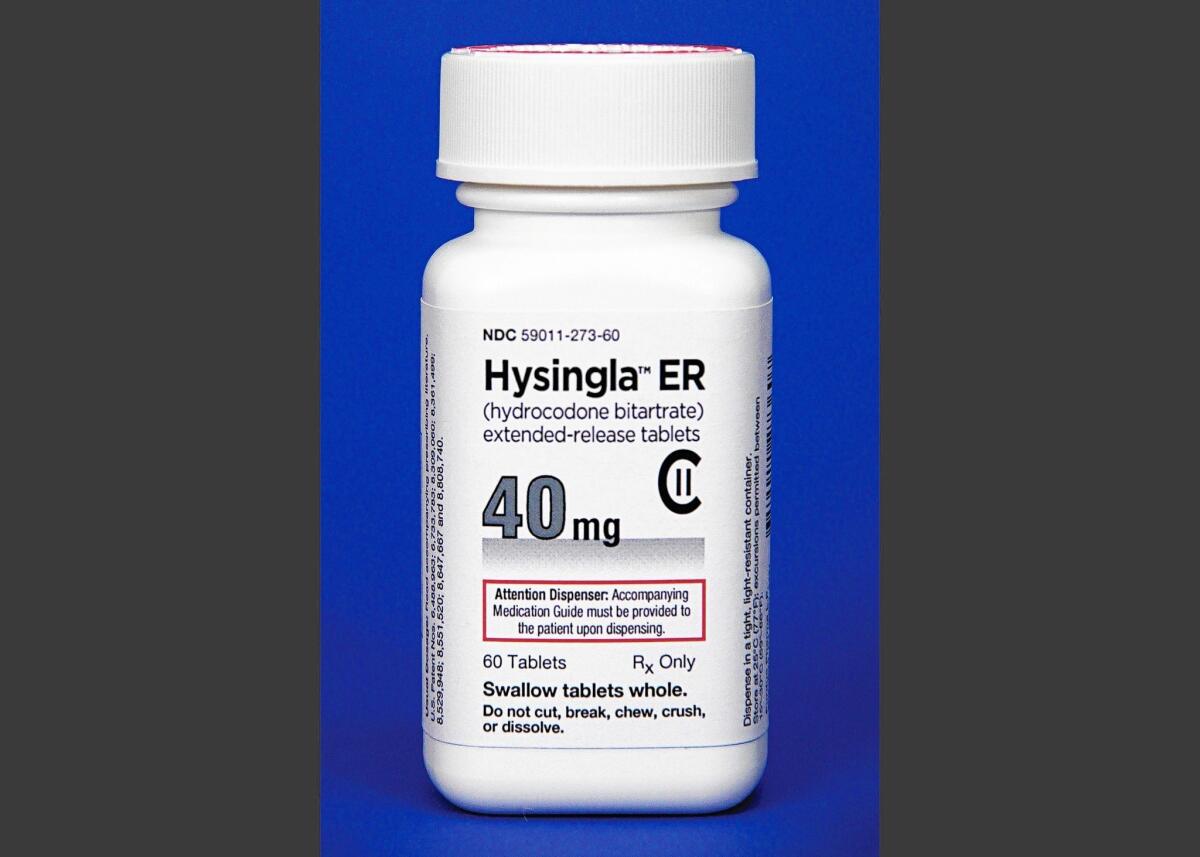
The U.S. Food and Drug Administration’s approval of Hysingla ER, an extended-release form of the widely prescribed hydrocodone, comes amid controversy over the proliferation of powerful narcotic painkillers in the face of an epidemic of addiction and overdose.
The maximum dose of Hysingla packs 120 milligrams of hydrocodone, the opioid analgesic contained in Vicodin, which is sold in 5- to 10-milligram doses. But when taken as directed, the new pill releases its medicine over 24 hours.
Hysingla is intended to be swallowed whole. The FDA expects it to reduce — but not totally prevent — people’s ability to abuse it by chewing it or smashing it into a powder that they snort or inject. The tablet forms a thick gel if it is crushed, making it difficult to inject.
But, the agency and the manufacturer acknowledged, abuse is still possible, and an overdose can result in death.
The approval of Hysingla is part of the FDA’s response to mounting overdose deaths involving prescription painkillers. The agency is encouraging drugmakers to make narcotic painkillers safer by developing abuse-deterrent formulations.
Dr. Janet Woodcock, director of the FDA’s Center for Drug Evaluation and Research, said Thursday’s approval of Hysingla was a move in the right direction.
“While the science of abuse deterrence is still evolving, the development of opioids that are harder to abuse is helpful in addressing the public health crisis of prescription drug abuse in the U.S.,” Woodcock said in a statement.
“Preventing prescription opioid abuse is a top public health priority for the FDA, and encouraging the development of opioids with abuse-deterrent properties is just one component of a broader approach to reducing abuse and misuse,” she said.
At the same time, Woodcock said, the agency wants to ensure “patients have access to appropriate treatments for pain.”
Hysingla’s only active ingredient is hydrocodone. Other forms of hydrocodone, such as Vicodin and Norco, also contain acetaminophen, which carries the risk of liver toxicity, the FDA noted in its review.
Mark Timney, chief executive of Purdue Pharma, said his company was proud to bring patients another pain treatment option. Hysingla is the company’s third approved abuse-deterrent opioid. The active ingredient in the others is oxycodone, a close cousin of hydrocodone.
“These innovations are an important step forward in helping meet patients’ needs while also working to deter misuse and abuse,” Timney said in a statement.
Hysingla comes a little more than a year after the FDA approved Zohydro ER, another long-acting form of hydrocodone. Designed to last 12 hours, Zohydro is available in strengths of up to 50 milligrams — 10 times the lowest dose of Vicodin. Because Zohydro can easily be crushed, its approval prompted criticism from addiction experts and public health officials who feared it would quickly become abused.
Dr. Andrew Kolodny, an addiction expert, said he feared that it was just a matter of time before addicts figure out how to abuse Hysingla.
“I think it’s an extremely dangerous product,” said Kolodny, who helped found Physicians for Responsible Opioid Prescribing. “A pill that’s so strong makes me very concerned that this product may be more dangerous than Zohydro.”
Purdue Vice President of Health Policy Dr. J. David Haddox said it was misleading to suggest that Hysingla is stronger than other painkillers.
“The range of tablet strengths for Hysingla ER is comparable to the strengths of several opioids already on the market, none of which have abuse-deterrent properties,” Haddox said in an email.
Opioid drugs — including forms of morphine, methadone, oxycodone and hydrocodone — are prescribed for patients who need powerful painkillers. But if patients take them for an extended period, they can develop a tolerance and require higher and higher doses of the drugs, increasing the risk of overdose and other problems.
Painkiller deaths quadrupled between 1999 and 2011, mirroring a sharp rise in the number of prescriptions for such drugs. In 2009, overdoses involving painkillers pushed drug fatalities past traffic accidents as a cause of death. And in 2011, the U.S. Centers for Disease Control and Prevention declared an epidemic.
The crisis had long been blamed on pharmacy robberies, teenage pill poppers and the black market. But a 2012 Los Angeles Times study showed that physicians played an important role in prescription drug overdoses. The Times analysis of 3,733 fatalities found that drugs prescribed to patients by physicians caused or contributed to nearly half the deaths.
The Hysingla approval did not immediately affect the FDA’s stance on Zohydro, but an agency spokesman said it would continue to watch for abuse. If a drug is determined to be unsafe, the agency can move to pull it off the market.
lisa.girion@latimes.com
Twitter: @lisagirion







