Curiosity rover’s findings on Mars’ organic molecules offer tantalizing clues in the search for life on the planet
- Share via
Over nearly six years roaming the surface of Mars, NASA’s Curiosity rover has detected organic molecules that offer a taste of what an ancient life-friendly Red Planet might have looked like. Now, the rover’s pulled together a veritable feast.
The discovery of a wide variety of organic molecules and the detection of a seasonal methane pattern — described in two separate studies in the journal Science — add new fuel to the search for past life on Mars, scientists said.
“Both these finding[s] are breakthroughs in astrobiology,” Inge Loes ten Kate of Utrecht University, who was not involved in either study, wrote in a commentary on the paper.
Ever since the twin Viking landers touched down in 1976, scientists have hunted for signs of organic molecules on Mars. At their most basic, organic molecules are those made of carbon and hydrogen atoms, though they can have other atoms (such as oxygen or sulfur) as well. They can come from living and nonliving sources, but because many are necessary for and produced by life on Earth, they’re considered possible biosignatures — signs of the presence of life.
Curiosity, which has been exploring Gale Crater since it landed in 2012, has previously dug up signs of organic molecules in the ancient lakebed, by taking rock samples and subjecting them to the suite of laboratory instruments in its belly. The problem was that these organic molecules contained an unusual atom: chlorine.
“The thing about the chlorinated molecules is that it’s not what you’d typically find in natural samples, and so we weren’t sure what the significance was at the time,” Jennifer Eigenbrode of NASA’s Goddard Space Flight Center in Maryland, lead author of one of the papers, said in a briefing. “However, it did give us a lot of motivation to keep looking.”
So the rover was directed to drive about 4 miles away to the base of Mt. Sharp, the 3-mile-high mound in the middle of Gale Crater whose layers of sediments serve as individual chapters in the geological history of Mars. Samples were drawn from two locations, Mojave and Confidence hills, and subjected to Curiosity’s Sample Analysis at Mars instrument suite.
For the previous mud stone samples that had produced the chlorinated molecules, scientists had heated the powdered rock to 200 degrees Celsius. But for this sample, Eigenbrode and her colleagues only analyzed the gases that were released above 400 degrees Celsius.
At that temperature, “they can be certain that these gases are not a result of leaking reagent or reaction with perchlorate,” Ten Kate wrote.
The results revealed a wealth of organics, Eigenbrode said — including some that had carbons linked in ring structures (such as benzenes) and others that include carbon chains (such as propane).
“Because we see these coming off of the sample at high temperatures, what they’re really telling us is that they’re part of something larger, a macromolecule,” she said.
Such a macromolecule might potentially look something like kerogen, Eigenbrode said — a substance that’s found in coal and black shale and meteorites. On Earth, it’s often the result of ancient plant or microbial matter.
These organic molecules had managed to survive in rock that was about 3.5 billion years old, and that may have lain within a few centimeters of the surface for perhaps 100,000 years, Eigenbrode said.
That may be because many of the compounds, such as thiophene, methanethiol and dimethyl sulfide, had sulfur atoms in their molecular structure — which would strengthen the relatively fragile organic molecules, allowing them to survive the radiation bombarding the planet’s surface for so long.
“There were a lot of people who thought we weren’t going to find all the organic molecules that we did,” Eigenbrode said. With this new, diverse set of molecules, “we can now start to understand a little bit more about how this material is preserved and where else we might look to get more.”
The second paper, led by Christopher Webster of Jet Propulsion Laboratory, marks a major step toward solving the mystery of methane on Mars. Methane is one of those key organic molecules that on Earth is largely produced by living things, and can also be eaten by them, and so could theoretically serve as a signal of the possibility or presence of life. But methane can also be produced by normal geologic processes.
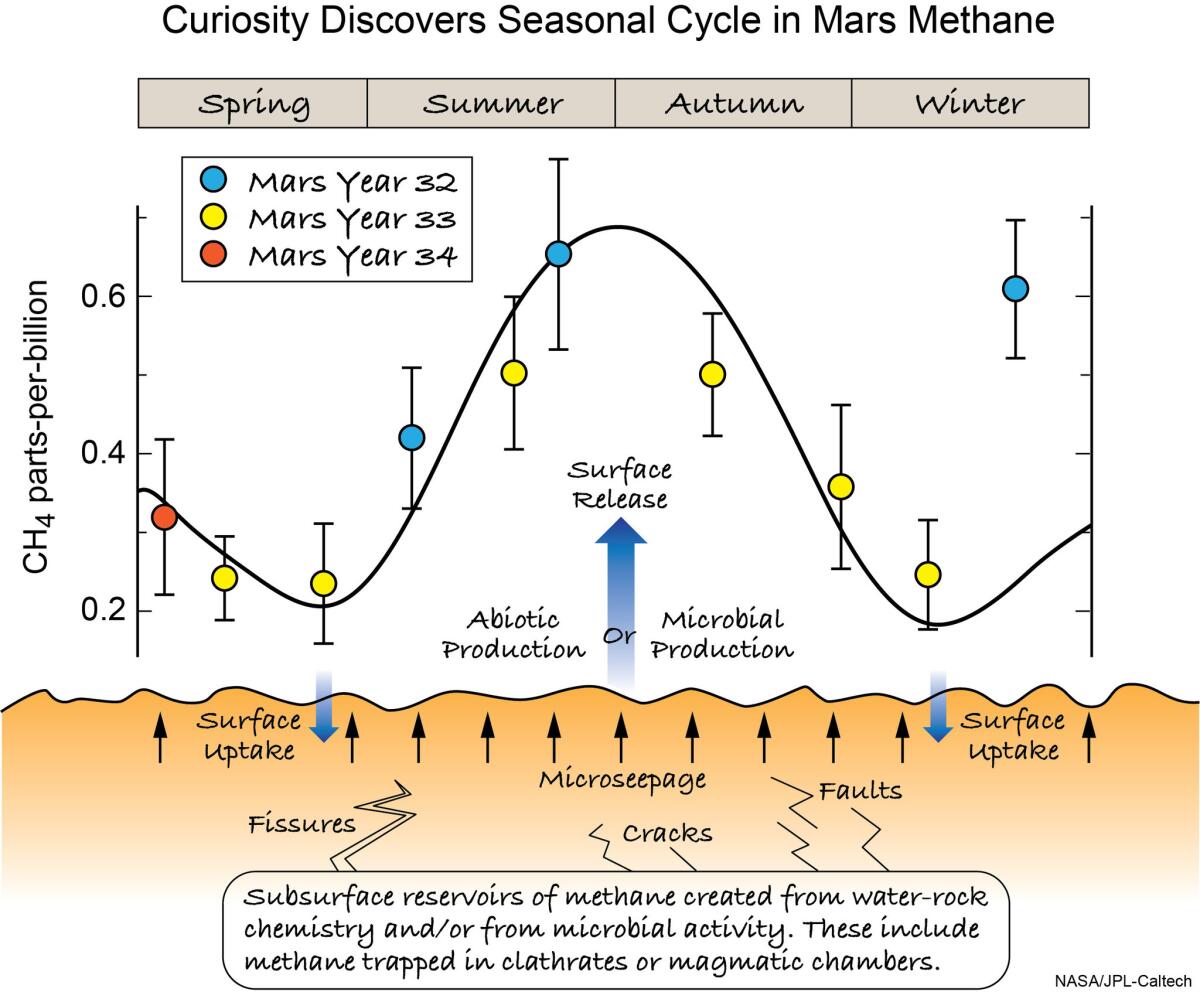
The first step to figuring out whether a methane signal is biological or not is to determine where it’s coming from. On Mars, that’s been a maddening challenge: While scientists have detected bursts of methane on the planet, they’ve appeared at random — and thus, it’s been difficult to figure out what the source is.
Now, with years of Curiosity’s atmospheric readings at their disposal, Webster and his colleagues were able to analyze 55 Earth months (or roughly three Martian years) of data, finding that there were indeed low levels of background radiation — and that it seemed to experience seasonal surges, nearly tripling at its peak near summer’s end in the northern hemisphere (and winter’s end in the south).
This seasonal pattern seems to imply that temperature changes might be triggering the seasonal release, the scientists said, suggesting that the methane might be stored in water-based crystals called clathrates.
“That’s a huge change, completely unexpected,” Webster said at the briefing. “And what it does is give us the key to unlocking the mysteries associated with methane, because now we have something to test our models and our understanding against.”
The source of that methane, however, remains a mystery, he said.
“We don’t know if that methane is ancient, we don’t know if it’s modern — it could be either,” Webster said. “We also don’t know if that methane was created from water-rock chemistry like serpentinization or if it was created by methanogen microbes. We cannot distinguish that.”
Neither paper can say whether past life ever existed on Mars, the scientists said. But they do provide new clues that could help researchers unravel this astrobiological mystery.
“The question of whether life might have originated or existed on Mars is a lot more opportune now that we know that organic molecules were present on its surface at that time,” Ten Kate wrote.
Follow @aminawrite on Twitter for more science news and "like" Los Angeles Times Science & Health on Facebook.
MORE IN SCIENCE
Dunes of methane ice? Tiny Pluto's surface reveals big surprises
Hurricanes and typhoons are slowing down, which means more time to do damage
On Thanksgiving, partisan politics cost Americans nearly 74 million hours with family and friends




