Global Development: As Ebola outbreak threatens nine African countries, new vaccine is main hope for preventing a major epidemic
- Share via
Reporting from Johannesburg, South Africa — Hopes of containing a growing outbreak of the Ebola virus in the Democratic Republic of Congo are pinned on an experimental vaccine that was tested in 2015 during a major outbreak of the disease.
The World Health Organization plans to vaccinate up to 10,000 people in the first phase of its response and so far has imported 4,000 doses of the vaccine, which is manufactured by the pharmaceutical giant Merck and appears to be highly effective in preventing infection.
The outbreak was first detected this month in a rural area known as Bikoro. The threat of the disease rapidly spreading grew with the discovery of four cases in Mbandaka, a city of1.2 million that lies on the Congo River.
The river is a major trade route linking Mbandaka to the capital, Kinshasa, as well as to Brazzaville, the capital of neighboring Republic of Congo.
An emergency meeting of the World Health Organization on Friday decided that the first doses of the vaccine will go to people who had contact with confirmed cases in Mbandaka, and the next will go to contacts of contacts. Vaccinations are expected to begin Sunday or Monday.
The organization stopped short of declaring the outbreak an international health emergency. But Dr. Robert Steffen, chairman of the WHO emergency committee, said nine countries in the region had been advised to prepare themselves for Ebola cases.

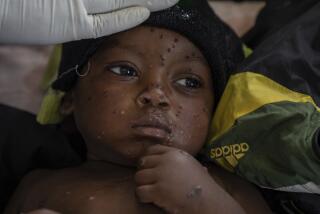
At least 45 people, including three medical workers, are thought to have been infected in the current outbreak, and 25 have died. In an effort to contain the spread, health workers have tracked down more than 500 people who had contact with people who were infected.
Of the four cases in Mbandaka, one had been confirmed by laboratory testing. The other three involve members of one family who came from a remote region and attended a funeral. The WHO was notified of the outbreak in Mbandaka on May 8.
Fighting Ebola in a crowded urban setting is deeply complicated.
Though the new vaccine proved effective in clinical trials in Guinea in 2015, it is difficult to trace people who had contact with the virus in a major city.
In the biggest outbreak of Ebola — when 28,000 people were infected and 11,300 died in the West African countries of Liberia, Sierra Leone and Guinea from 2013 to 2016 — the virus spread swiftly to cities from a rural trade hub near the borders of the three nations.
A recent outbreak of bubonic plague in Madagascar underscored the difficulty of trying to contain epidemics in cities.
Normally limited to a remote mountainous area of the country, the bacterial disease spread to the capital, Antananarivo, and other cities after one man contracted it in the hinterlands and took a commuter minibus through the capital to the coast.
What might have been a few dozen cases became 2,348, causing 202 deaths.
In the case of the Ebola infection confirmed in Mbandaka, Steffen said it was not clear how it was contracted.
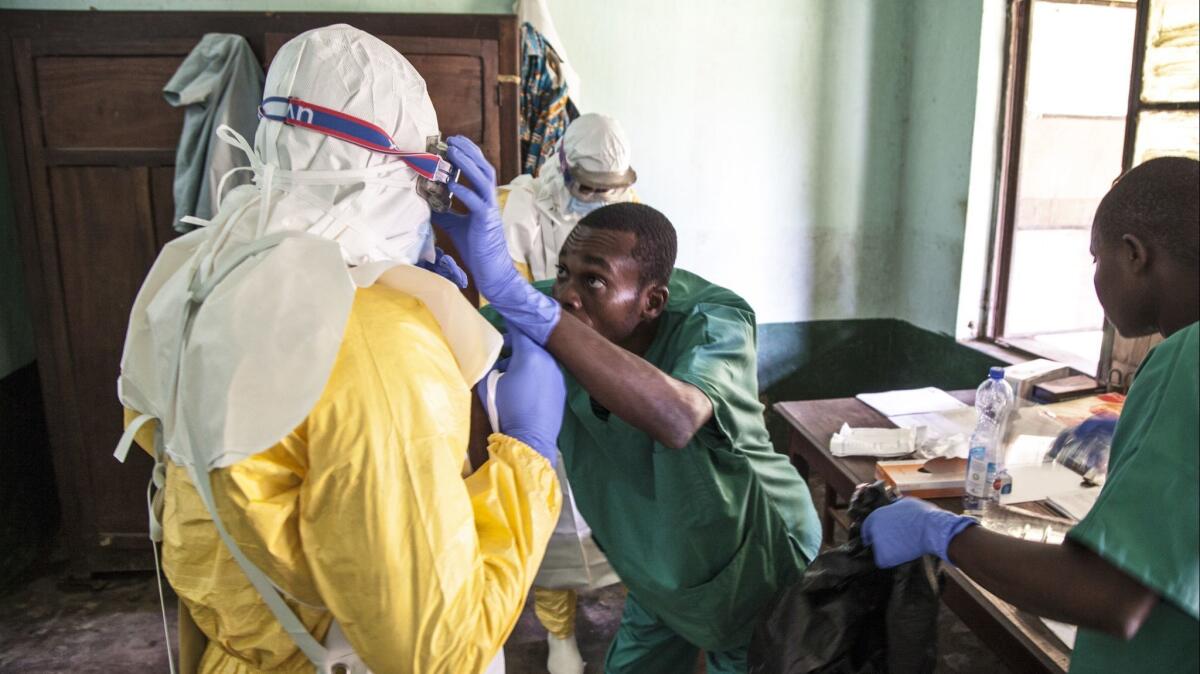
Until that case was found, the outbreak had been concentrated in remote villages near Bikoro, a market town on the shores of Lake Tumba. Many of the villages in the area are accessible only along narrow dirt tracks on foot or motorcycle. WHO experts who responded to the crisis last weekend took a helicopter.
Steffen said authorities now face the complex task of monitoring dozens of small ports along the Congo River for possible cases. Thirty WHO staffers have been deployed to the region, along with members of the health organization Doctors Without Borders, which played a major role in fighting the 2013-16 outbreak.
At the outset of that epidemic, the available treatment was limited to rehydration. Victims were crowded into isolation wards in an effort to stop the spread of the virus, but in many cases intravenous fluids were not provided because using needles was seen as too risky for medical staff.
Dying patients were simply given bottled water, but many were too sick to drink it.
Westerners who were evacuated to hospitals in Europe and the U.S. fared better than the West Africans. Locals often hoped their symptoms were from less-fatal diseases such as malaria or typhoid and put off checking into Ebola treatment units, from where few survivors emerged.
By the time they went for treatment, it was difficult to save them.
This time, WHO experts are optimistic that those who suspect they may have the illness will report to Ebola treatment facilities early, trusting that the vaccine could save their lives.
In the Guinea trial, 5,837 people who had been in contact with someone who had the virus — or someone who had been in contact with the contact — got vaccinations. None of them became infected with the virus, according to a study published in the Lancet medical journal.
The vaccine was believed to have played a role in ending the 2013-16 outbreak.
The vaccine has not yet been licensed to treat humans, but Congolese officials authorized its use. This is the first time it will be used to try to prevent an outbreak from spreading in its early stages.
One potential challenge: The vaccine has to be stored between minus-76 and minus-112 degrees Fahrenheit, which is difficult in a remote region where electricity is unreliable.
The WHO has sought $26 million from donors over three months to cover the vaccination campaign. The 2013-16 outbreak cost the international community as much as $4 billion.
The Ebola virus was discovered in 1976, and except for the major outbreak in Liberia, Sierra Leone and Guinea, it has mainly been found in rural villages in central Africa. Because those outbreaks were relatively easy to contain, little work was done to develop a vaccine.
Some research took place shortly after Sept. 11, 2001, amid fears that the virus could be weaponized in a terrorist attack. The effort laid the groundwork for the vaccine that was tested in Guinea.
Twitter: @RobynDixon_LAT
More to Read
Sign up for Essential California
The most important California stories and recommendations in your inbox every morning.
You may occasionally receive promotional content from the Los Angeles Times.