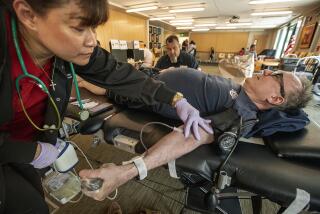
Panel considers lifting FDA ban on blood donations by gay men
- Share via
A U.S. Department of Health and Human Services panel has begun to consider whether to overturn a long-standing ban against accepting blood donations from gay men.
On Thursday, the HHS Advisory Committee on Blood and Tissue Safety and Availability heard testimony from advocates who say that the lifetime ban is discriminatory and that technological advances have made it obsolete.
------------
FOR THE RECORD
Nov. 14, 9:04 a.m.: An earlier version of this post stated that the Advisory Committee on Blood and Tissue Safety and Availability was a Food and Drug Administration panel. It is a panel of the Department of Health and Human Services.
------------
“The ban on gay and bisexual men ... was enacted in 1985 and focuses on sexual orientation more than the risk and science itself,” said Caleb Laieski, a 19-year-old gay activist who has sued the Food and Drug Administration to overturn the ban.
“A recent study by the American Red Cross estimates that lifting the blood donation ban could be used to help save the lives of more than 1.8 million people,” Laieski told the group.
The ban in the United States applies to any prospective male blood donor who has had sex with another man since 1977, the beginning of the AIDS epidemic in the U.S., the FDA explains on its website.
The FDA said the ban was necessary because gay and bisexual men are at an increased risk for HIV, hepatitis B and other infections that can be transmitted via blood transfusion.
“HIV tests currently in use are highly accurate, but still cannot detect HIV 100% of the time,” the FDA states on its site. “It is estimated that the HIV risk from a unit of blood has been reduced to about 1 per 2 million in the USA, almost exclusively from so-called ‘window’ period donations.”
The window period exists very early after infection, when viral levels are too low to be detected by current methods. “For this reason, a person could test negative, even when they are actually HIV positive and infectious,” the FDA says.
Other industrialized countries, including Australia, Britain, France, Italy and Japan, have overturned similar bans on blood donation. Instead of barring gay and bisexual men for life, some of the countries allow them to donate blood if they have not had sex with another man in the last 12 months.
Laieski is not alone in opposing the U.S. ban. Last year, the American Medical Assn. voted to oppose the FDA policy, calling it “discriminatory and not based on sound science.”
In addition, three groups that supply nearly all of the nation’s blood — the American Red Cross, AABB and America’s Blood Centers — have long advocated ending the prohibition.
Follow @montemorin for science news