Dermatologist Barred From Injecting Liquid Silicone : Courts: Judge rejects contention that treatment for acne scarring and wrinkles is exempt from U.S. regulation. He orders the chemical destroyed.
- Share via
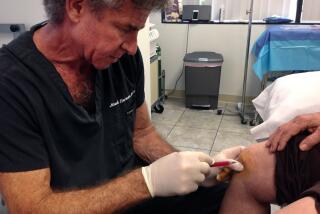
LOS ANGELES — A federal judge has barred a Newport Beach dermatologist from injecting liquid silicone in the faces of his patients, rejecting the physician’s contention that this widespread treatment for acne scarring and wrinkles is exempt from government regulation.
U.S. District Judge William J. Rea also ordered Dr. James E. Fulton Jr. to destroy, under the supervision of the U.S. Food and Drug Administration, all of the liquid silicone in his possession. Fulton was given 10 days from last Wednesday’s entry of the order to dispose of his inventory of the outlawed substance at his own cost.
Liquid silicone is an illegal product that is not approved by the FDA for any uses. Nonetheless, it has been used by dermatologists and plastic surgeons nationwide for smoothing acne blemishes and wrinkles.
Essentially, the material is a liquid form of the gel used in once-popular silicone breast implants that the FDA earlier this year banned from common use.
In the 1970s, liquid silicone was studied as a potential treatment for facial deformities but the research was dropped after complications developed, such as redness, sores and other serious side effects. Nonetheless it continued to be used by many physicians.
FDA Commissioner David A. Kessler has said he intends to crack down on the medical use of illegal products, specifically naming liquid silicone. He said Wednesday he was gratified by the judge’s decision.
“This case should send a signal to both the medical community as well as patients that the use of this product is illegal, dangerous and will not be tolerated,” he said.
Neither Fulton nor his lawyer, Jay H. Geller, could be reached for comment late Wednesday. Connie Largent, the doctor’s office manager, said only that Fulton currently is not using silicone injections in his practice.
Federal authorities filed suit Feb. 28 against Fulton, co-developer of Retin-A, a derivative of Vitamin A that has shown some effectiveness as a topical anti-wrinkle and anti-aging treatment. The suit also named the Acne Research Institute, a nonprofit research foundation, and Vivant Inc., both of Newport Beach.
More to Read
Sign up for Essential California
The most important California stories and recommendations in your inbox every morning.
You may occasionally receive promotional content from the Los Angeles Times.