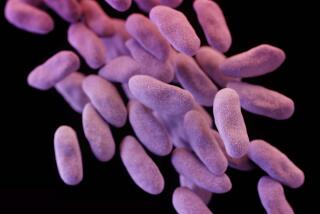
A mysterious adversary preys on the war’s wounded
- Share via
The young Army medic would not stop bleeding.
He had been put on a powerful regimen of antibiotics by doctors aboard the hospital ship Comfort in the Persian Gulf. But something was wrong.
He was in shock and bleeding from small pricks where nurses had placed intravenous lines. Red, swollen tissue from an active bacterial infection was expanding around his abdominal wound. His immune system was in overdrive.
How odd, thought Dr. Kyle Petersen, an infectious disease specialist. He knew of one injured Iraqi man with similar symptoms and a few days later encountered an Iraqi teenager with gunshot wounds in the same condition.
Within a few days, blood tests confirmed that the medic and the two wounded Iraqis were all infected with an unusual bacterium, Acinetobacter baumannii.
This particular strain had a deadly twist. It was resistant to a dozen antibiotics. The medic survived, but by the time Petersen connected the dots, the two Iraqi patients were dead.
It was April 2003, early in the Iraq war -- and 4 1/2 years later, scientists are still struggling to understand the medical mystery.
The three cases aboard the Comfort were the first of a stubborn outbreak that has spread to at least five other American military hospitals, including Walter Reed Army Medical Center in Washington and the Army’s Landstuhl Regional Medical Center in Germany.
Hundreds of patients -- the military says it has not tabulated how many -- have been infected with the bacterium in their bloodstream, cerebrospinal fluid, bones or lungs. Many of them were troops wounded in Iraq or Afghanistan; others have been civilians infected after stays in military hospitals.
At least 27 people have died in military hospitals with Acinetobacter infections since 2003, although doctors are uncertain how many of the deaths were caused by the bacteria.
The rise in infections has been dramatic. In 2001 and 2002, Acinetobacter infections made up about 2% of admissions at the specialized burn unit at Brooke Army Medical Center in Texas. In 2003, the rate jumped to 6%, and then to 12% by 2005. Other military hospitals have reported similar increases.
In the early days of the war, there were so many infections in an intensive care unit on the Comfort that a nurse posted a sign: “Acinetobacter Alley.” In two months, the bacterium was found in 44 of the 211 patients wounded in battle.
It was getting out of control. Petersen pleaded for help on an infectious disease mailing list.
“Can anyone familiar with [the] soil biology of Iraq or the drug-prescribing practices of the pre-regime medical system explain the severe drug resistance pattern we are seeing among our trauma victims?”
A persistent slacker
It was no surprise that Petersen knew little about Acinetobacter -- which has long been seen as the slacker of the bacterial world.
It’s called Acinetobacter, from the Latin word for “motionless,” because the bacterium lacks flagella or cilia to move.
“Organisms that are relatively wimpy pathogens . . . are not high on people’s list,” said Fred Tenover, a microbiologist at the Centers for Disease Control and Prevention in Atlanta.
The bacterium is persistent, however, and requires few nutrients. It lives naturally in soil and can survive for days on dry surfaces, such as doorknobs or hospital equipment.
Acinetobacter usually threatens only the weakest of the weak, those whose immune systems are compromised because of old age, trauma or disease. Even then, garden-variety Acinetobacter is easily controlled with common antibiotics.
But the situation started to change about two decades ago.
Acinetobacter followed an evolutionary path trod by numerous other bacteria since World War II, when antibiotics were introduced widely. Bacteria not killed by an antibiotic would pass on their resistance to later generations.
The process was quickened by the often profligate use of the drugs, which allowed more bacteria to develop resistance.
Today a host of diseases, such as tuberculosis and gonorrhea, have highly antibiotic-resistant strains.
“If we use antibiotics to kill off everything else, what is left standing is very, very drug resistant,” said CDC epidemiologist Arjun Srinivasan. “Acinetobacter is one of those left standing.”
Tenover first noticed a strain of Acinetobacter with some drug resistance in the mid-1980s while working at a veterans hospital in Seattle. Several years later, he met with Ghassan Matar, a visiting Lebanese scientist at the CDC, whose samples of Acinetobacter baumannii from patients in a Beirut hospital raised another red flag.
The infections were a legacy of years of fighting. Positive tests for Acinetobacter more than tripled at the hospital from 1983 to 1984 and stayed high for years after. The samples Matar brought were already resistant to two important classes of antibiotics.
In the following years, civilian hospitals in the U.S. and around the world reported sporadic outbreaks of drug-resistant Acinetobacter.
“You have an organism of relatively low virulence that became more important because you’ve run out of drugs to treat it with,” Tenover said.
The question that Petersen struggled with was how this bug had found its way into modern military hospitals.
Doctors could beat back an infection with the strongest antibiotics, and hospitals could try to scrub away the bacteria. But those weren’t solutions.
They had to find the source of the contamination.
The plea is heard
Responses to Petersen’s plea on the mailing list poured in to the Comfort.
A Canadian soil scientist who worked in Iraq in the 1970s described high rates of antibiotic-resistant Staphylococcus bacteria in dirt samples.
The scientist surmised they were caused by the erratic distribution of antibiotics in Iraq. A shipment of drugs would arrive and doctors would use them until they ran out. Then they would prescribe whatever other antibiotic was sent next, the scientist said.
A microbiologist wrote to Petersen about Australian patients injured in the 2002 nightclub bombings in Bali, Indonesia, who returned home with astronomically high levels of very drug-resistant bacteria, including Acinetobacter.
“It gave me an idea that maybe it was something related to the process of aeromedical evacuation or the injury process,” said Petersen, now 39.
After Comfort reported its first Acinetobacter cases, infections sprang up in military hospitals in the Middle East, Germany and the U.S. The facilities took the cases seriously.
The night Marine Maj. K.C. Schuring arrived at Andrews Air Force Base in Maryland, a doctor told him point-blank that if his fever didn’t subside within three days, his left leg would probably have to be amputated.
Schuring, barely conscious and lying on a gurney, heard the doctor tell him that the infection could spread: “This can kill you.”
He was taken to the National Naval Medical Center in Bethesda, Md., and immediately isolated. He heard the word Acinetobacter for the first time.
Schuring, who had been shot in both legs in Iraq, could take bad news, but this worried him.
“I was happy they could treat it, though they said they couldn’t necessarily cure it,” he said.
Whenever he left the room, he wore a yellow gown to alert others of his infection. Everyone who visited him donned yellow gowns and gloves.
He felt “like a freak,” he said.
Digging up dirt
In 2003, Dr. Clint Murray, then a 33-year-old Army major at a frontline aid station in Iraq, began to dig for answers.
He started at the beginning.
Though some wounded soldiers were sent to aid stations such as Murray’s, most were airlifted directly to more advanced facilities like the Combat Support Hospital in Baghdad.
Murray, an infectious disease specialist, asked a critical care doctor there to take samples from soldiers wounded by guns, improvised explosive devices, mortars or other weapons. As doctors scrambled to stabilize patients, the wounds were swabbed to collect bacteria. Most of the samples were taken within 20 to 40 minutes of the soldiers’ injuries.
But out of the samples taken from 49 patients, the doctors found no Acinetobacter, though there were plenty of other bacteria, such as Staphylococcus.
That still left the possibility that dirt and dust from beyond the battlefield had blown into a wound.
Murray joined a group, including Petersen and Srinivasan, that focused on dirt around field hospitals in Iraq and Kuwait -- the next step in the medical evacuation chain that started at frontline aid stations and ended at hospitals in the U.S.
The group gathered 18 dirt samples around seven field hospitals, and also looked at 31 archived soil samples from around the combat zone.
Only one of the soil samples -- taken from outside a field hospital mess hall -- turned up positive. The group compared it with strains collected from casualties at the field hospital in Baghdad and larger hospitals including Landstuhl in Germany, and found they were not related.
Dirt, it seemed, was not the culprit.
In late 2004, Murray returned to Brooke Army Medical Center. He wondered whether soldiers were carrying the bacterium on their skin and infecting themselves when wounded. He set to work on a study swabbing the nostrils of 293 soldiers at Ft. Sam Houston, Texas, who had never been to Iraq or Afghanistan.
None of the soldiers tested positive for Acinetobacter, Murray and colleagues reported in the journal Infection Control and Hospital Epidemiology in 2006.
They seemed to be running up against a wall.
As the search continued, military doctors struggled to find an effective strategy to combat the bug. The treatment could be difficult.
Schuring said doctors experimented with different drugs. Schuring’s situation was complicated by his allergy to penicillin.
At one point, he was taking four antibiotics. The infection had already taken away his appetite and made him queasy. It took doctors about two weeks to narrow down his treatment to a relatively new intravenous antibiotic, tigecycline.
Doctors operated on him nearly every other day to clean out dead tissue in his legs.
When the infection began to settle down, doctors installed a 21-inch stainless steel plate along his left thigh. The surgery made Schuring dizzy, but the doctors didn’t want to give him a blood transfusion for fear of inciting a new infection.
The treatment, Schuring said, was like going “through hell.”
Suspicion shifts
Evidence was building that the cause of the infections was something in the military trauma system.
The hospital-based transmission made sense since the bacterium had already taken up residence in civilian hospitals.
Though preliminary typing found no link between the U.S. civilian strains and the military casualty strains, the conditions in military hospitals were just right for the bacterium, said Srinivasan, the CDC epidemiologist.
In the hectic environment of field hospitals, it was also difficult to impose strict infection-control measures, such as thorough cleaning of hands and equipment after each patient, Murray said.
The field hospitals had become the center of a perfect storm of trauma -- battle- hardened bugs preying on the weakest patients at their most vulnerable moments.
“Soldiers now survive injuries they wouldn’t have survived before,” Srinivasan said.. “That challenge creates soldiers who are sick, who are living in healthcare facilities for a long time and are more susceptible to hospital-associated infections.”
It took about three years for Murray and his colleagues to look through the entire chain of trauma, from the battlefields to the field hospitals to the tertiary care center in Landstuhl and finally to the military hospitals in the U.S.
The results of their labor, published this May in the journal Clinical Infectious Diseases, showed that all seven field hospitals tested in Iraq and Kuwait had Acinetobacter in patient care areas.
“We can’t be 100% sure, but the data supports that patients are probably getting exposed to Acinetobacter in field hospitals in Iraq,” Murray said.
How the bacteria became entrenched in the field hospitals is still unknown. But in many ways, it is irrelevant. It is there, and, as civilian hospitals have also found, it is not going away easily.
The military strain of the bacterium has caused at least one civilian death. Acinetobacter was growing in the lungs and bloodstream of a 35-year-old man whose immune system was suppressed because he had a kidney transplant at Walter Reed in 2005. There were no signs of infection until the man came down with acute shortness of breath one evening and died soon after.
For the most part, doctors have figured out the most effective drugs against the bacterium -- an antibiotic called imipenem and an older class of drugs known as polymyxins. The drugs have made the infections fairly manageable. Through stricter controls, such as monitored hand-washing, infection rates have shown signs of dropping in some hospitals.
Petersen, who worked at National Naval Medical Center in Bethesda after the Comfort’s mission ended, treated just one or two Acinetobacter infections in July, down from the highs of 15 to 20 a month in 2004 and 2005. This year, there have been only a few cases each month, according to hospital figures.
But Murray now wonders whether Acinetobacter was really the culprit after all.
He and others looked at patients with the worst outcomes at Brooke’s burn unit and found that Acinetobacter was associated with larger burns but was not causing more deaths by itself.
A study of 35 returning soldiers with the most extreme kinds of shin bone fractures found that Acinetobacter was the most common bacterium around fracture sites when the patients arrived, but it was easy to clear.
Those who later suffered serious complications, including amputations, tended to be infected with other serious bacteria, such as Staphylococcus and Pseudomonas aeruginosa.
Acinetobacter, it turns out, may only be a marker of vulnerability. “It is not the worst bug,” Murray said.
‘One step at a time’
The battle between bacteria and humans never ends.
Recently, scientists have noted signs that Acinetobacter strains are growing resistant to polymyxins and imipenem, said Tenover, the CDC microbiologist.
There are, however, small victories for humans.
Just before Christmas, after a month of treatment, Schuring returned to his home in Farmington Hills, Mich.
Schuring’s wife, Lynn, was nervous about this strange bug her husband had brought back from the war.
What if they kissed? What if her husband put down a drink and one of their young children took a sip? Would her parents, who are in their 70s, be at risk if they visited?
Doctors assured her that this bug was no danger to the strong. So far, no one in the family has gotten sick.
But Schuring’s doctor warned them that they must keep an eye out for any sign of the bacteria, which could lie dormant for years.
Schuring, now a 38-year-old lieutenant colonel, has continued to improve and hobbles around on his own two legs. He has one last surgery at Bethesda in January and then, perhaps, he will run marathons again.
“You know, this is a long process for these guys and their families,” Lynn Schuring said. “I think we just take it one step at a time. . . .
“Everything we’ve been through has taught me to take it one step at a time.”
More to Read
Sign up for Essential California
The most important California stories and recommendations in your inbox every morning.
You may occasionally receive promotional content from the Los Angeles Times.