Smallpox vaccine used in risky cancer therapy seized from San Diego stem cell company

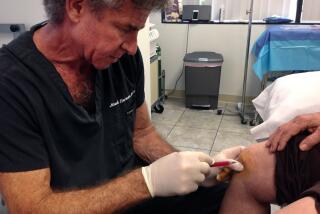
Five vials of the smallpox vaccine were seized from StemImmune by the U.S. Marshals Service on behalf of the FDA.
- Share via
Cancer patients were given an unapproved and potentially dangerous treatment from a San Diego stem cell company that contained smallpox vaccine, the U.S. Food and Drug Administration said Monday.
The FDA announced a broader nationwide crackdown on “unscrupulous clinics” using illicit treatments after federal agents took action against San Diego’s StemImmune.
“I will not allow these activities to go unchecked,” FDA Commissioner Scott Gottlieb said in a statement.
Five vials of the vaccine were seized from StemImmune by the U.S. Marshals Service on behalf of the FDA. The vials, one partially used and four intact, originally contained 100 doses each of vaccine.
The action and an FDA warning given to a Florida stem cell clinic were welcomed by researchers who had warned for years of dangers from a proliferation of unregulated stem cell clinics.
The vaccine contains live vaccinia virus, related to cowpox and smallpox. It’s safe when given to healthy people. But since vaccination eradicated smallpox in nature decades ago, the vaccine is no longer used except for those at risk to exposure to bioweapons.
California Stem Cell Treatment Centers gave the vaccine-containing treatment to cancer patients with potentially compromised immune systems at its locations in Rancho Mirage and Beverly Hills, the FDA said in a statement.
Recent inspections at both StemImmune and California Stem Cell Treatment Centers confirmed the vaccine was combined with fat-derived cells called stromal vascular fraction to create the unapproved treatment, the agency said.
“The unproven and potentially dangerous treatment was being injected intravenously and directly into patients’ tumors,” the FDA said.
The vaccine is not commercially available, the agency said, so it has “serious concerns” about how StemImmune got the vaccine.
StemImmune was called and emailed for comment Monday morning, but as of early Monday afternoon has not replied.
Also Monday, the FDA posted a warning letter to US Stem Cell Clinic of Sunrise, Fla., and its Chief Scientific Officer Kristin Comella for “marketing stem cell products without FDA approval and for significant deviations from current good manufacturing practice requirements, including some that could impact the sterility of their products, putting patients at risk.”
Three elderly patients with macular degeneration were left blind after getting treatment from the company, according to an article in the New England Journal of Medicine. Stem cells were extracted their from fat, mixed with blood plasma and injected into their eyes.
The actions are part of a new FDA policy to ensure proper oversight of stem cell clinics, Commissioner Gottlieb said in a statement. Gottlieb was confirmed as commissioner earlier this year after being nominated by President Donald Trump.
“I’ve directed the FDA to launch a new working group to pursue unscrupulous clinics through whatever legally enforceable means are necessary to protect the public health,” Gottlieb said in his policy statement.
Hundreds of stem cell clinics offering unapproved or questionable therapies operate around the country, according to a 2016 study published in the journal Cell Stem Cell.
While these clinics have proliferated in recent years, federal oversight has been lax, the study said. Unauthorized treatments are even being marketed on a web site operated by the National Institutes of Health, according to another study published earlier this year in the journal Regenerative Medicine.
The FDA’s response after years of lax enforcement is encouraging, said bioethicist Leigh Turner, a co-author of the 2016 study and author of the newer study.
“I think it’s a striking development,” said Turner, associate professor at the University of Minnesota Center for Bioethics. “This seems to be a sign the FDA is increasing enforcement in this area, dedicating resources, putting together a working group. And then we have two examples of action being taken.”
Stem cell researcher Paul Knoepfler, the other co-author of the 2016 study, likewise praised the FDA’s stepped-up oversight of stem cell therapy.
“This is pretty historic,” said Knoepfler, a professor at UC Davis School of Medicine’s Department of Cell Biology and Human Anatomy. “We’ve never really seen strong action and words from the FDA, all at once.”
The FDA said any harm from treatments at California Stem Cell Treatment Centers should be reported to the agency’s MedWatch Adverse Event Reporting program.
To file a report, use the MedWatch Online Voluntary Reporting Form, available at bit.ly/medwatchfda. The completed form can be submitted online or faxed to 1-800-FDA-0178.
For further reading
Statement from FDA Commissioner Scott Gottlieb
Dubious stem cell clinical trials marketed on NIH website
Unapproved stem cell therapies multiply
Science Playlist


In a first, scientists rid human embryos of a potentially fatal gene mutation by editing their DNA

10 interesting facts about Mars

Kids can add years to your life
LA 90: SpaceX launches recycled rocket

Ocean temperatures warming at rapid rate, study finds
bradley.fikes@sduniontribune.com
(619) 293-1020
More to Read
Inside the business of entertainment
The Wide Shot brings you news, analysis and insights on everything from streaming wars to production — and what it all means for the future.
You may occasionally receive promotional content from the Los Angeles Times.