Subterranean bacteria are prepared to survive antibiotics
- Share via
No place on Earth demonstrates the resilience or inventiveness of life quite like Lechuguilla Cave, whose subterranean tunnels stretch for 130 miles through Carlsbad Caverns National Park in New Mexico.
Deep in the cave’s most arid recesses, deprived of all sunlight and mostly starved of life-giving water, a lush garden of bacteria grows. Untouched by humans for all of their 4 million years, these strains of bacteria thrive on the harsh minerals of the geological formations to which they cling and fend off other life forms that would prey on them.
It is a simple life. But new research suggests it could tell us volumes about the medicines doctors rely upon to combat infection and why, increasingly, they are failing.
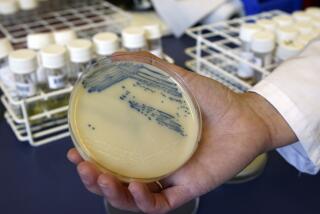
Scientists who collected 93 strains of bacteria from the forbidding depths of Lechuguilla found that all were resistant to at least one of the antibiotics that modern medicine uses to fight bacterial infections and some were resistant to at least 14. In addition, virtually all of the 26 antibiotics tested as part of the study proved useless in killing at least one of the strains of bacteria collected.
That these life forms evolved in ways that appear to anticipate medicines attests to bacteria’s remarkable powers of survival. It also suggests that the rise in antibiotic- resistant diseases isn’t due entirely to the runaway use of these drugs; rather, try as you might to kill them, bacteria are programmed to endure.
“It’s awe-inspiring,” said Gerard D. Wright, a microbiologist from the Institute for Infectious Disease Research at McMaster University in Canada and senior author of the study, which was published online Wednesday by the journal PLoS ONE. “It gives you real respect for the genetic diversity and the ability of these organisms to evade toxic molecules.”
The study appeared on the same day that the Food and Drug Administration announced it would ask drug makers and veterinarians to drastically reduce the widespread use of antibiotics to promote the growth of commercial livestock. And it suggests that while such measures may slow the rate at which infectious diseases gain the upper hand against medicines, they cannot stop that process.
Scientists have long believed that the ability of disease-causing bacteria to outwit antibiotic medicines was a man-made phenomenon, said Eileen Choffnes, director of the Institute of Medicine’s forum on microbial threats. The growing use of antibiotics derived from plants and synthesized in laboratories was thought to have spurred adaptations that made many of these bacterial pathogens less vulnerable to drugs used to fight tuberculosis, malaria, gonorrhea, influenza, pneumonia and AIDS.
But the new research demonstrates that antibiotic resistance emerged millions of years before those medicines were used — and in an environment far too forbidding for the bacteria to have come into any contact with the drugs, Choffnes said.
The findings make it clear that humans will always have to contend with the problem of antibiotic resistance, no matter what steps are taken to prevent it, said Dr. Brad Spellberg, an infectious disease researcher at the Los Angeles Biomedical Research Institute at Harbor-UCLA Medical Center: “There’s never going to be a point where we can say, ‘OK, we’re up, we’re ahead, they’re done.’ ”
Researchers have harvested bacteria on the Earth’s surface that was thousands of years old and reported last year that some of them were resistant to antibiotics. That might have been due to exposure to natural antibiotic threats in their environment.
“This pushes it way back,” Spellberg said.
For the new study, about 500 strains of bacteria from three sites in the cave were brought to the surface by Hazel A. Barton, a spelunker and microbiologist at the University of Akron in Ohio. To harvest them, she burrowed into areas of the cave where the National Park Service could ensure that no more than six humans had ever been near.
Barton’s samples were kept alive in lab dishes that approximated their nutrient-poor origins. The 93 strains that survived and were chosen for evaluation were subjected to 26 antimicrobial agents, ranging from natural products such as vancomycin to completely synthetic agents such as ciprofloxacin and linezolid.
In one group of bacterial strains — the “gram-positive” strains — 70% of the samples were resistant to between three and four classes of antibiotics, on average. The same was true of 65% of the “gram-negative” strains.
Tetracycline antibiotics were effective against all of the bacterial samples. But sulfamethoxazole, trimethoprim and fosfomycin were not. Three ancient strains of bacteria in the Streptomyces genus proved resistant to daptomycin, the newest class of antibiotic approved for clinical use.
In an interview, Barton said that the discovery that ancient bacteria were immune to so many modern medicines was “a eureka moment.”
“As a scientist, that’s what you live for,” said Barton, who combined a childhood passion for caving with her microbiology research. “Just like in cave exploration, it’s the discovery of the unexpected that keeps you going back for more.”
Barton added that the study’s findings should help scientists developing antibiotics for future use to anticipate ways in which bacteria might adapt to resist the effects of new medicines.
Spellberg said the findings underscore the need for measures like the one taken Wednesday by the FDA. In 2010, the agency said that nearly 29 million pounds of antibiotic agents were fed to the nation’s livestock each year. That practice accelerates bacteria’s adaptation to the drugs, he said.
But Spellberg added that the federal government should take a more activist role in ensuring that the development of new antibiotic agents — a risky and expensive enterprise that has scared off many pharmaceutical companies — continues. The Generating Antibiotic Incentives Now (GAIN) Act, a bill now pending in Congress, would create economic incentives for drug companies to invest in developing new antibiotics, he said.