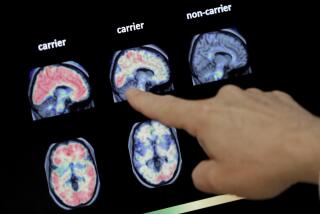
2 new drugs aim to prevent migraines; early tests done
- Share via
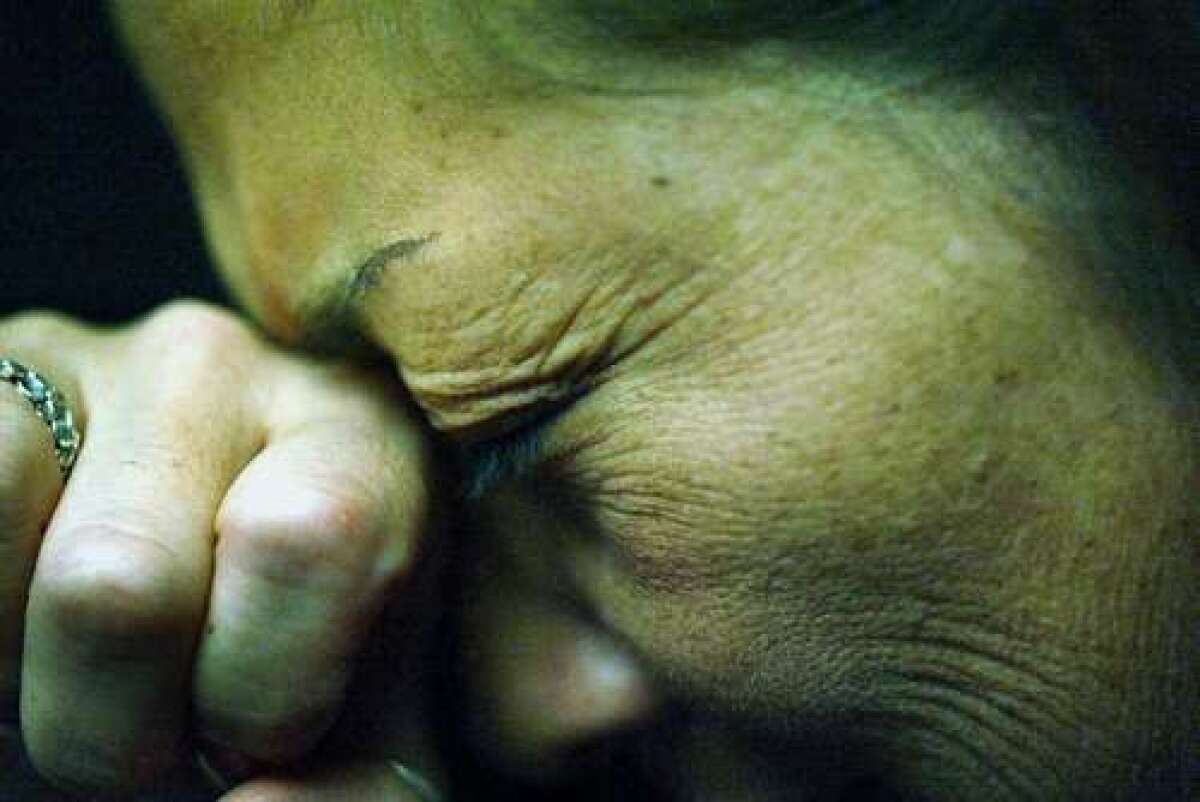
Two drugs given to people who suffer migraines reduced the frequency of their headaches in early trials, scientists said.
The test results “may potentially represent a new era in preventive therapy for migraine,” Dr. Peter Goadsby, an author on studies of both drugs, said in a statement. One of the researchers called migraine headaches the third most common medical disorder in the world.
Both drugs must undergo larger trials to confirm the results.
Both drugs are intended to prevent rather than treat migraine headaches, and the studies of them are the first to test monoclonal antibodies for migraine prevention, the scientists said. The drugs are directed against what’s called the calcitonin gene-related peptide, which had been thought important in migraines but had not previously been targeted with a drug.
The researchers are to present their findings at the American Academy of Neurology’s 66th annual meeting, which begins Saturday in Philadelphia.
“The big deal is that there’s never been anything introduced to prevent migraine attacks that was based on a mechanism and a sole indication of migraine,” Goadsby said Wednesday by telephone. In the past, sufferers could try beta blockers, antidepressants or anticonvulsants, hoping they’d work for their headaches.
“This the first bespoke migraine treatment,” said Goadsby, a neurology professor at Kings College London and UC San Francisco.
He said patients should be pleased, and for some it could be life-changing. He gave the example of a pilot who could be unable to keep flying if her headaches were treated with certain drugs now in use.
In one study 163 people who had migraines from five 14 days a month were given a placebo or an intravenous dose of a drug called ALD403. They were followed for 24 weeks. Those who got the drug had an average of 5.6 fewer migraine days a month; those who took the placebo had 4.6 fewer days.
In the second study, 217 people who had a migraine four to 14 days a month, received biweekly injections of a drug called LY2951742 or a placebo for 12 weeks. Those who got the drug had an average of 4.2 fewer migraine days a month at the 12-week mark. Those who got the placebo had an average of three fewer days.
Dr. David Dodick of the Mayo Clinic in Arizona, an author of both studies, said by telephone that rates for placebos are often high in studies of pain, and in this case those rates could be due in part to the high level of anticipation people had for the success of migraine treatment. They also could be affected, as they sometimes are, by the invasiveness of the treatments – injections rather than pills, he said.
“Clearly in the future, we’ll have to get a grip on this and carefully, carefully design” the next phase of trials for the drugs, he said.
The scientists don’t have a sense yet of which drug might be better, or might work better for certain people, Dodick said. Both appear safe at this point, he said.
The studies were supported by drug companies, ALD403 by Alder Biopharmaceuticals and LY2951742 by Arteaus.
Twitter: @mmacvean