Europe frets over slowdown in COVID-19 vaccine supply

- Share via
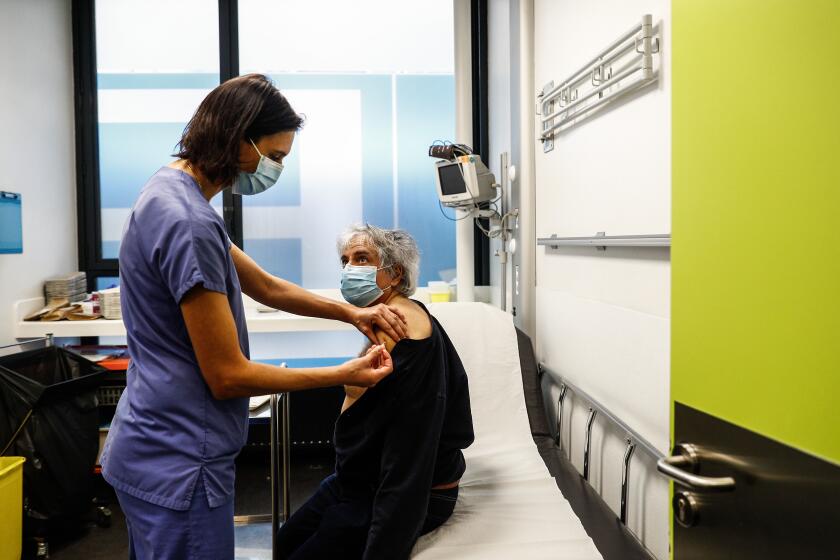
BRUSSELS — The European Union is pressuring drugmaker AstraZeneca to deliver more COVID-19 vaccine doses to its 27 nations and to stick to its initial supply promises once the shot receives EU approval, especially since the bloc has already invested in enhancing production capacity.
EU Commission President Ursula von der Leyen held urgent talks Monday with AstraZeneca chief Pascal Soriot, and EU nations are also meeting with the British-Swedish company to press it to increase its vaccine production and meet its contractual targets.
The talks follow AstraZeneca’s announcement that it would be dispatching fewer doses of its vaccine than anticipated early on in its delivery schedule once the shot received its expected authorization from the European medicines regulator later this week. The announcement came as a blow on top of Pfizer’s failure last week to keep up promised deliveries of its own COVID-19 vaccine, which the company says are being reduced temporarily so that it can revamp its plant in Belgium to increase overall production. Italy has threatened to sue Pfizer for the delays.
Despite the economic and political clout that comes with being the biggest trading bloc in the world, the EU is lagging badly behind countries such as the U.S., Israel and Britain in the rollout of COVID-19 vaccines for its most vulnerable population and for healthcare workers. The bloc’s leaders have faced strong criticism for moving so slowly.
The European Medicines Agency is scheduled to review the Oxford-AstraZeneca coronavirus vaccine Friday, and its approval is hotly anticipated. The vaccine is already being used in Britain and has been approved for emergency use in half a dozen countries, including India, Pakistan, Argentina and Mexico.
Political pressure on EU leaders spurred the European Commission into action Monday, with Von der Leyen’s phone call to the AstraZeneca chief.
The French are among the most reluctant people in the world to get a COVID-19 shot because of distrust of the government and past health scandals.
“She made it clear that she expects AstraZeneca to deliver on the contractual arrangements foreseen in the advance purchasing agreement,” said Von der Leyen’s spokesman, Eric Mamer.
“She reminded Mr. Soriot that the EU has invested significant amounts in the company up front precisely to ensure that production is ramped up even before the conditional market authorization is delivered by the European Medicines Agency.”
Mamer acknowledged that “production issues can appear with the complex vaccine, but we expect the company to find solutions and to exploit all possible flexibilities to deliver swiftly.”
The European Commission also wants a transparency register to record and approve all export of vaccines out of the EU to third countries.
“We, as the EU, must be able to know whether and what vaccines are being exported from the EU,” German Health Minister Jens Spahn said. “Only that way can we understand whether our EU contracts with the producers are being served fairly. An obligation to get approval for vaccine exports on the EU level makes sense.”
The delays in receiving doses are likely to make it difficult for the EU to meet early targets in its plan to vaccinate 70% of its adult population by late summer.
EU Council President Charles Michel said the EU already “pounded our fist on the table” with Pfizer last week to ensure that Pfizer’s delivery delays ended by the end of this week.
The EU has signed six vaccine contracts for more than 2 billion doses, but only the Pfizer-BioNTech and Moderna vaccines have been approved for use so far in the bloc’s 27 nations.
More to Read
Sign up for Essential California
The most important California stories and recommendations in your inbox every morning.
You may occasionally receive promotional content from the Los Angeles Times.