Pfizer begins studying effects of a 3rd COVID-19 vaccine dose
- Share via
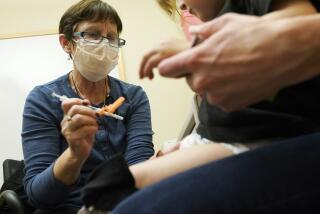
NEW YORK — Pfizer announced Thursday that it had begun studying the effects of a third dose of its COVID-19 vaccine, part of a strategy to guard against mutated versions of the coronavirus.
Health authorities say first-generation COVID-19 vaccines still protect against the variants that are emerging in different parts of the world. But manufacturers are starting to prepare now in case a more vaccine-resistant strain comes along.
Pfizer said it would offer a third dose to 144 volunteers, drawing from people who participated in the vaccine’s early-stage U.S. testing last year. The company wants to determine if an additional booster shot given six to 12 months after the first two doses will rev up the immune system enough to ward off a mutated virus.
Pfizer and its German partner, BioNTech, also are tweaking their vaccine recipe, which in essence instructs the body to build copies of the spiked proteins found on the coronavirus. The companies are in discussions with U.S. and European regulators about a study to evaluate doses updated to better match coronavirus variants such as the one first discovered in South Africa.
More to Read
Sign up for Essential California
The most important California stories and recommendations in your inbox every morning.
You may occasionally receive promotional content from the Los Angeles Times.