Sanofi and GSK say their COVID-19 vaccine strongly protects adults in early trials

- Share via
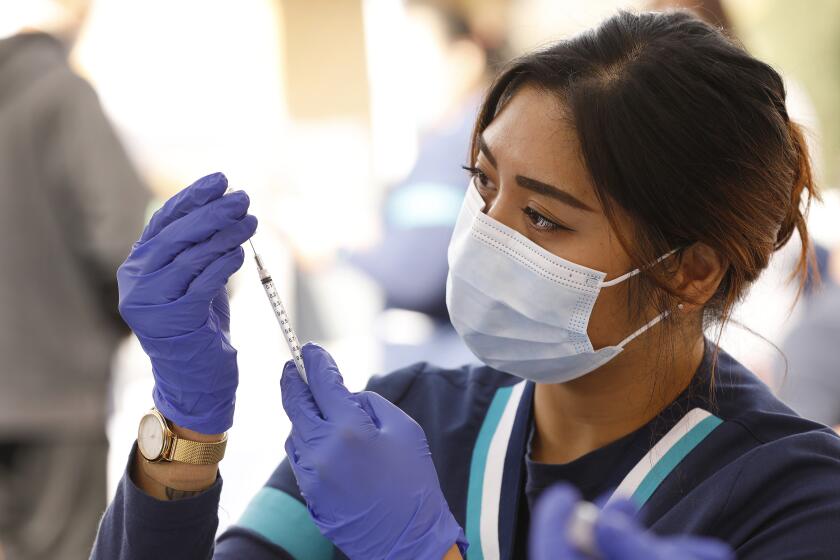
PARIS — Sanofi and GlaxoSmithKline’s potential COVID-19 vaccine triggered strong immune responses in all adult age groups in preliminary trials, boosting hope that the shot could join the fight against the pandemic this year.
After two doses of the vaccine candidate, participants showed neutralizing antibodies in line with those found in people who had recovered from the disease, according to results of the Phase 2 trial released Monday. The drugmakers said they plan to begin late-stage trials and production in the coming weeks and hope to win regulatory approval for the vaccine before the end of 2021.
Regulators around the world have already authorized a number of COVID-19 vaccines, such as those by Pfizer-BioNTech, Moderna, AstraZeneca and Johnson & Johnson. But experts say more are needed as public health authorities race to vaccinate people amid a pandemic that has already killed more than 3.3 million people and caused economic havoc.
The Sanofi-GSK vaccine was an important part of the European Union’s vaccination strategy and had been championed by French President Emmanuel Macron’s government. But researchers had to reformulate it after early testing produced an inadequate immune response in older people.
L.A. officials also reported Sunday that 325 people were in the hospital with COVID-19, a significant drop from a peak of more than 8,000 hospitalized COVID-19 patients in January during the region’s winter surge.
The Sanofi-GSK candidate joins about a dozen vaccines now undergoing late-stage trials. The companies plan to produce up to 1 billion doses annually, and they have signed agreements to supply the U.S., Canada and developing countries. Public health experts say several vaccines will be needed to end the pandemic, because of the challenges in rapidly producing and distributing enough doses to vaccinate billions of people.
“We know multiple vaccines will be needed, especially as variants continue to emerge and the need for effective and booster vaccines which can be stored at normal temperatures increases,” said Thomas Triomphe, head of Sanofi’s vaccines unit.
The results released Monday were from a Phase 2 trial involving 722 volunteers aged 18 to 95 who were recruited in the U.S. and Honduras.
The late-stage trial will involve about 37,000 participants from countries around the world, the companies said.
More to Read
Sign up for Essential California
The most important California stories and recommendations in your inbox every morning.
You may occasionally receive promotional content from the Los Angeles Times.