Stem cells created without destroying embryos
- Share via
Scientists reported Thursday that for the first time they have made human embryonic stem cells without destroying embryos, a development that the government’s top stem cell official said would make the controversial research eligible for federal funding.
Story Landis, who chairs the National Institute of Health’s stem cell task force, said that with certain safeguards, the new method appeared to comply with federal restrictions that have largely cut scientists off from the $28 billion the government spends on medical research each year.
Federal law prohibits the National Institutes of Health from paying for experiments that place human embryos at risk of injury or death, and spending on human embryonic stem cell research is restricted to projects involving a handful of cell lines that were created before August 2001.
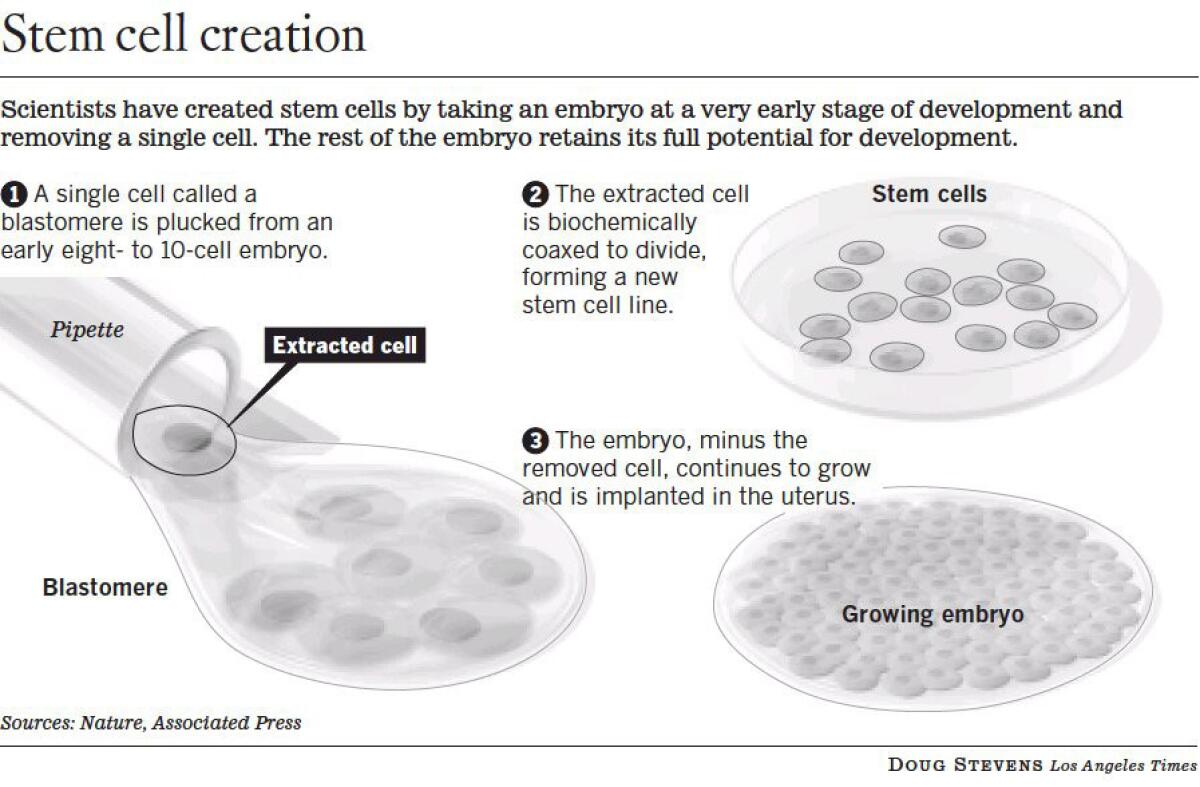
Researchers at Advanced Cell Technology in Worcester, Mass., created four stem cell lines out of individual cells plucked from 3-day-old embryos, which continued to develop normally after the procedure. The method was described in the online edition of the journal Cell Stem Cell.
The removal of a single cell from a young embryo is done thousands of times a year in the U.S. by fertility laboratories to screen embryos for genetic diseases.
Dr. Robert Lanza, Advanced Cell Technology’s chief scientific officer, said researchers could piggyback on the procedure by allowing the removed cell to divide in a laboratory dish. Then, with the consent of patients, one copy could be used for genetic screening and the other to make stem cells.
Under those circumstances, the research “should be permissible under the Bush policy,” said Russell Korobkin, a stem cell expert at the UCLA School of Law. “The NIH should not decline to fund this.”
Though the technique spares embryos, it still raises ethical concerns.
The Rev. Tadeusz Pacholczyk, an ethicist at the National Catholic Bioethics Center in Philadelphia, said that removing a single cell from an embryo turns it into “a starting source for harvestable raw materials, in a gesture that reduces young humans to commodities.”
And because the single embryonic cell can be grown into stem cells, some scientists and ethicists wondered whether the cell itself has the potential to become a whole new embryo.
“It would be hard to rule out,” said Martin Pera, director of the Center for Stem Cell and Regenerative Medicine at USC.
New optimism
But for scientists who have struggled under federal restrictions, the technique has injected a sense of optimism that there may soon be a break in the political logjam that scientists blame for stalling progress in the field.
After all, the idea of producing stem cells by taking a single cell from an embryo was suggested by the President’s Council on Bioethics in a 2005 white paper that examined ways to make the valued cell lines without creating, harming or destroying embryos.
“The president’s own bioethics advisory council suggested this approach,” said Sean Morrison, director of the University of Michigan Center for Stem Cell Biology, who was not involved in the study. “The administration has a responsibility to now support it.”
Michigan is one of at least nine states where it is illegal to derive new human embryonic stem cell lines using traditional methods, even with private funding. Morrison said the new method appears to comply with Michigan law.
The study puts the finishing touches on a highly touted experiment 19 months ago that demonstrated the feasibility of the process but did not actually preserve any embryos, largely for practical reasons.
The approach relies on culling individual embryonic cells using a biopsy technique known as pre-implantation genetic diagnosis, or PGD. About 5,000 couples in the U.S. opt for PGD each year, according to the Society for Assisted Reproductive Technology.
In late 2005, Lanza demonstrated that the technique was possible in mice. The donor embryos were implanted into surrogate mothers and resulted in live births 49% of the time, virtually the same as for intact embryos.
The following year, Lanza’s team showed that a single cell taken from an early-stage human embryo could be grown into a stem cell line. To maximize their chances of success, they used all of the cells, called blastomeres, from the donor embryos and therefore couldn’t track whether their development would have been hampered by the biopsy.
In the latest study, the researchers used 41 embryos that were frozen by fertility clinics. They thawed them and prompted them to grow to the eight-cell stage, then used a tiny glass pipette to remove one or two blastomeres. Most of the donor embryos continued to develop normally for two more days before they were refrozen.
The individual cells were grown in dishes and surrounded by human embryonic stem cells and other compounds that induced them to develop into embryonic stem cells.
The scientists created four cell lines that were able to grow into all the main tissue types in the body, including neurons and beating heart cells.
Colleagues at UC San Francisco repeated the experiment with two more embryos, but they did not use established stem cells to help the blastomeres grow. They produced a stem cell line that didn’t involve any materials derived from dismantled embryos.
There is a potential flaw with the technique: Even the delicate removal of one cell could place an embryo’s health at risk.
“How do you demonstrate that the embryo was not harmed?” Landis said. “Right there, you have a significant problem.”
Protecting embryo health
The ideal experiment -- in which embryos that have been biopsied to make stem cells are implanted in surrogates so that the babies can be studied for medical problems -- is impossible to conduct for health and ethical reasons.
But Lanza said there is a straightforward solution.
To avoid potential risk to embryos, scientists could piggyback on PGD biopsies that clinics are performing anyway, he said. After the blastomere is removed, it could be nurtured in a dish for about 12 hours until it divides. Then one cell could be used for genetic screening and the other would be available for research.
Landis agreed that stem cells derived in this manner would appear to meet the federal standards.
Lanza said he believed that the method was safe enough to use on spare embryos from fertility clinics and still qualify for federal funding. In his experiments, 80% to 85% of the donor embryos developed to the stage where they could be implanted in a uterus -- a higher rate than at fertility clinics.
“Any clinic would be thrilled with 85%,” said Dr. Mark Hughes, director of the Genesis Genetics Institute in Detroit. “Many clinics would be happy with 60%.”
Landis said there was no way to know whether the embryos that didn’t survive were lost because of the initial thawing or the subsequent biopsy.
Researchers could get some idea by comparing the pregnancy rates of couples that used PGD to those of couples that opted for standard in vitro fertilization.
A study published last year in the New England Journal of Medicine found that among older women who had IVF, those who also used PGD were about 30% less likely to have a baby. But specialists who perform the diagnostic procedure said that the study was flawed and that an alternative reading of the data showed that genetic screening slightly boosted the odds of having a baby.
The entire exercise of trying to satisfy the Bush policy could become moot after the presidential election if a new administration scraps the restrictions. But Lanza said the researchers couldn’t wait.
“We need more lines to study,” he said. “We can’t afford to waste time.”







