Study tests if a warm heart makes a better transplant
- Share via
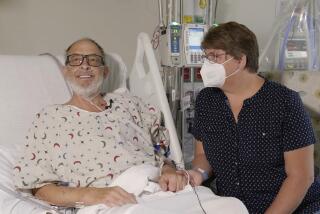
Rob Evans, a 61-year-old social worker from Apache Junction, Ariz., got the good news on Father’s Day: After 31/2 years, doctors had found him a heart and were preparing to bring it to UCLA, where he was being treated for a slow, steady decay of his cardiac muscle. Evans had been hospitalized at UCLA for six weeks.
Excited, hopeful and anxious all at once, Evans dared imagine a different life: out in the garage remodeling his ’69 Nova, riding his horse, wrestling with his grandson and helping his wife, Gail, take care of their barn instead of sitting, exhausted, in his chair all day.
FOR THE RECORD:
Doctor’s name: An earlier version of this article misspelled the first name of Dr. Bartley Griffith, the director of heart and lung transplantation at the University of Maryland in Baltimore, as Bartely.
But the heart destined for Evans’ chest was not like most that transplant recipients get. Instead of coming to him in icy slumber inside a cooler, it was warm and beating. Though it had been excised from its donor, blood flowed through it until shortly before it was stitched into Evans’ body on June 19.
Evans is taking part in a nationwide clinical trial to test whether such “beating hearts” are as good as, or better than, hearts packed in ice and slowed down with potassium and other chemicals. Doctors at UCLA, one of the study’s leading sites, have enrolled multiple patients, and other transplant centers across the country will soon start recruiting too.
“The human heart was never meant to be on ice,” said Dr. Abbas Ardehali, the surgical director of UCLA’s heart and lung transplantation program and the trial’s principal investigator. Chilling donor hearts before a transplant slows the rate of cell death and buys time to get the organ to a recipient, but only a little bit, he said. “That’s why having a human heart in a warm, beating state has always been exciting.”
It lives
From the outside, it looks like an ordinary gray-and-white storage unit on wheels. At 40 inches tall, 30 inches wide and 20 inches deep, it weighs less than 100 pounds and fits comfortably in the back of an SUV.
But inside, it houses a living human heart.
The beating organ rests in a clear, sterile chamber equipped with a pacemaker in case it needs a jump-start. Warm blood flows through tubing connected to the aorta because the veins that normally bring blood to the heart are sewn shut. The blood travels through the heart, feeding it oxygen and nutrients so it can function while removing waste products, which are filtered out by a machine. Fresh oxygen and nutrients are added to the donor’s blood before it is pumped back in. All the while, doctors can keep tabs on the heart’s rhythm, blood pressure and other vital signs to make sure it is healthy for surgery.
“The heart believes it’s still in the body,” said Dr. Waleed Hassanein, who founded TransMedics, the Massachusetts-based medical device company that developed the apparatus and is sponsoring the trial.
The technology was tested in Europe in a 2006 clinical trial of 20 heart transplant patients. Nineteen of these patients were out of intensive care within 24 hours, a much shorter stay than the two to five days patients usually spend in the ICU, said Dr. Bruce Rosengard, the surgical director of cardiac transplantation at Massachusetts General Hospital in Boston and one of the first surgeons to perform warm transplants in Europe.
“That’s a testament that those hearts function quite well,” he said. The device, known as the Organ Care System, has been commercially available in Europe since 2009.
Worldwide, more than 100 warm transplants have now been performed, all of them with positive results, Hassanein said. In the U.S., 24 patients have received warm transplants, including Evans.
To be sure, transplants of hearts packed in ice are lifesavers: More than 2,400 such hearts were transplanted in the U.S. last year, according to data from the United Network for Organ Sharing, the nonprofit organization in Richmond, Va., that oversees the country’s transplant system. Almost 90% of recipients survive the first year after surgery, and close to 50% live for at least 10 years with their new hearts, according to the National Heart, Lung and Blood Institute.
But there are drawbacks. Once a heart is disconnected from its blood supply, surgeons cool it to put the brakes on damage that occurs because cells are deprived of oxygen and nutrients. Damage still occurs in spite of the cooling, though — so much so that after about six to eight hours outside a body, the hearts must be thrown away. As a result, a heart in Los Angeles, for example, can’t be used to save a matched patient in Miami because the heart would be too debilitated by the time it arrived at the hospital for transplant.
In addition, surgeons only have time to use factors such as blood type, weight, height and gender to pair up donors and recipients. They don’t have the luxury of running more sophisticated tests, like they do for kidney transplants, because the heart won’t tolerate the delay.
“If we could test for proteins that mount an immune response, it could improve outcomes for transplant patients,” Ardehali said.
Cardiologists and cardiac surgeons are hoping the Organ Care System will give them more time to get hearts to the most critical — and best-matched — patients. The technology could also help them “buff up” hearts that may be slightly injured by the effects of brain damage, which could increase the number of available hearts, said Dr. Bartley Griffith, the director of heart and lung transplantation at the University of Maryland in Baltimore who was involved in a safety trial of the Organ Care System.
“That’s really the holy grail,” he said.
But first they have to test it head-to-head against the conventional transplant method.
To do so, they will enroll a total of 128 patients at UCLA and more than 10 other sites across the country. Doctors will assess how many patients survive 30 days after the surgery, how much time they spend in intensive care and how often they reject their new hearts. They anticipate being done with the trial by the end of 2012.
Conditions such as the time the heart is outside the body and the types of donors and recipients will be kept as similar as possible. The primary difference will be whether a patient gets a warm or a cold heart — and that won’t be known until the moment the transplant team gets ready to collect the organ.
At that time, a member of the team will open a sealed envelope.
If the piece of paper inside says “standard of care,” the team takes a regular cooler to the hospital where the heart is waiting. If the form says “OCS,” for Organ Care System, they bring the TransMedics device and nutrients for the heart.
One year ago, the sheet for Andrea Ybarra read “OCS.” She had been waiting for a heart for about 11/2 years.
The 41-year-old former daycare owner from Whittier went to the emergency room in 2003 thinking she had gallstones. She was tired, had lost her appetite and had pain in her back and abdomen. Her doctor sent her to a cardiologist, and she was diagnosed with cardiomyopathy, the same condition that Evans had.
In 2006, surgeons inserted a pacemaker. But by 2007, the pacemaker wasn’t enough. Ybarra was so tired and weak that she would fall asleep at the breakfast table. And multiple times she felt her pacemaker jolt her heart into beating again.
When she went to UCLA for treatment in 2007, doctors also diagnosed her with lupus and told her the autoimmune disorder had likely caused the damage to her heart.
She needed a heart transplant.
She joined the transplant list in 2008 and was told on three occasions that there might be a match for her, only to have those organs fall through.
“It was so frustrating. I felt like I was living on eggshells,” she said.
Then nurse practitioner Chris Eisenring, who is coordinating the warm-heart trial at UCLA, suggested she enroll. Ybarra and her husband, Moises, discussed it with their family and agreed to participate.
Today, Ybarra walks for 45 minutes on her treadmill six times a week to keep her new heart healthy. The routine biopsies (to make sure her body isn’t rejecting the heart) and the sonograms (to check that it’s working properly) are almost behind her.
She and her husband recently went on a road trip to Las Vegas.
“We’re starting slow,” she said.
The couple would like to have a baby someday.
“That will be my biggest accomplishment,” she said.
Evans is also making strides. Five weeks after his June 19 surgery, he walked up a hill. “It’s been 31/2 years since he’s been able to do anything like that,” said his wife, Gail.
Such dramatic improvements are to be seen whichever way a heart ends up in someone’s chest. It’s too early to say whether patients do better in the long run with hearts kept warm and beating.
But Evans is already convinced. “Fresh is always better than frozen,” he said while sitting on a bench outside a hotel in Westwood, where he stayed after he was healthy enough to be discharged from the hospital but not yet cleared to return to his home in suburban Phoenix.
He said he was eager to get back to his office at Mountain Health & Wellness, a nonprofit healthcare provider where he is chief executive. He also was looking forward to shooting hoops with his grandson and taking his motorcycle for a spin.
He has been back in Arizona since Aug. 16. Next week, he plans to go horseback riding for the first time in four years.