With Roe overruled, can red states disregard FDA rules on abortion pills?
- Share via
WASHINGTON — When the Supreme Court overturned Roe vs. Wade last month, many abortion rights advocates looked to pills sent by mail as a possible option for pregnant women who live in states where abortion is outlawed.
More than 20 years ago, the Food and Drug Administration approved abortion medication as safe and effective for ending an early pregnancy. And last year, partly in response to the pandemic, the agency said the pills may be distributed by a pharmacist or sent directly to a patient’s home by courier or by mail.
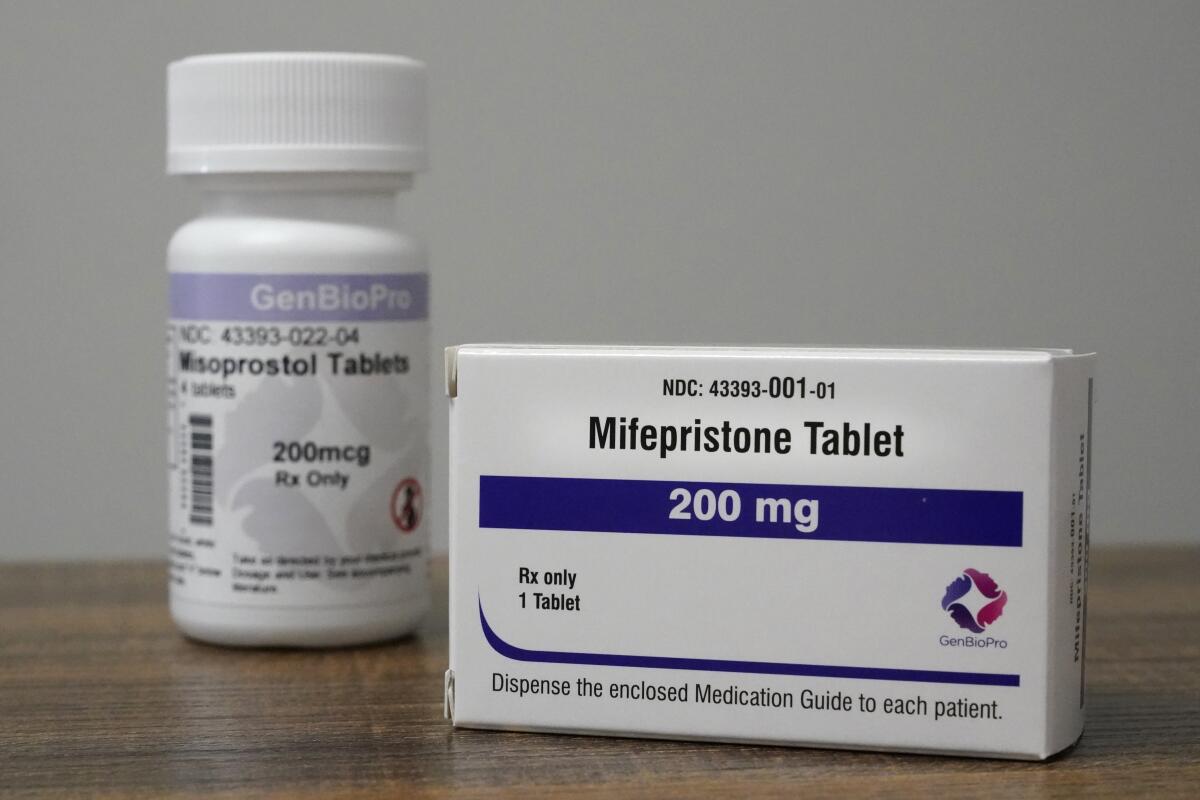
In recent years, two drugs taken in combination — mifepristone and misoprostol — became the most common form of abortion in the U.S., overtaking in-clinic procedures.
But antiabortion states questioned the safety of these drugs and restricted their use, even before the right to abortion was overturned. Texas, for example, made it a crime last year for anyone to send or deliver an “abortion-inducing drug” to a pregnant woman.
The federal-versus-state dispute has set the stage for another legal battle that could shape the rights of women in about half the nation.
At issue is whether the federal regulations on safe drugs could override state laws and, if so, provide a legal option for women where abortion is banned.
In response to the court’s ruling on June 24, President Biden and Atty. Gen. Merrick Garland said they would fight to defend the FDA rules.
“States may not ban mifepristone based on disagreement with the FDA’s expert judgment about its safety and efficacy,” Garland said.
A first case testing the issue is pending in Mississippi. It is one of 19 states that required a physician to see the patient when the abortion medication was prescribed, and it also said the patient must be “in same room” with the physician when she takes the pills.
Two years ago, lawyers for GenBioPro, the Nevada-based maker of a generic version of mifepristone, filed suit in federal court in Mississippi contending that its restrictions were extreme and pre-empted or overridden by federal law. They argued the Constitution makes federal law supreme, and they called the state’s rules “an impermissible effort by Mississippi to establish its own drug approval policy.”
U.S. District Judge Henry Wingate had put off ruling on the GenBioPro case until the Supreme Court decided the other Mississippi case — Dobbs vs. Jackson Women’s Health — which resulted in the June 24 ruling overturning Roe. Now his decision in GenBioPro vs. Dobbs could be the first on whether federal drug law means abortion pills may be legally distributed in antiabortion states.
“We should have a decision soon, but we will also be filing suits in other states,” said Ken Parsigian, a Boston lawyer who represents GenBioPro. “Right now, we have the only suit out there, and we’d love to have the Justice Department join us.”
The state’s lawyers called for the drugmaker’s suit to be dismissed. They said the Supreme Court gave the state “sweeping authority” to prohibit abortion, and that includes any “medicine or drug” that would terminate a pregnancy.
Legal experts said both sides in this dispute can claim the law in on their side.
“Congress intended the FDA to be the gatekeeper. States don’t get to supersede the FDA and deny the use of approved drugs,” said Temple University law professor Rachel Rebouché. But she said the state also has a strong argument that “it is regulating the practice of medicine,” not setting drug policy.
She and others said they were not aware of a state prohibiting the use of a drug approved by the FDA.
Congress did not say directly that federal drug laws always preempt extra restrictions imposed by a state, and the Supreme Court in the past has been divided on the question.
Twice in recent years, the justices ruled for a maker of a generic drug who sought to be shielded from state restrictions. At issue then was whether a drugmaker could be sued under state law and held liable for a patient’s horrible injuries because the FDA-approved label did not go far enough to warn of the danger.
The court’s conservatives, in a pair of 5-4 decisions, ruled the federal drug laws prevailed over the state’s extra restrictions.
“Under the Supremacy Clause, from which our pre-emption doctrine is derived, any state law, however clearly within a state’s acknowledged power, which interferes with or is contrary to federal law, must yield,” Justice Samuel A. Alito Jr. wrote in Mutual Pharmaceutical vs. Bartlett. A lower court had ruled the manufacturer could avoid the problem by not selling the drug in a particular state, but Alito said, “We reject this stop-selling rationale as incompatible with our pre-emption jurisprudence.”
But Alito and other court conservatives, having just upheld the state’s power to prohibit abortion, are probably the least likely to uphold the broad reach of the federal drug laws when states are blocking the use of abortion medication.
The Justice Department has not said whether it intends to join the court fight to defend the FDA’s rules.
Greer Donley, a University of Pittsburgh law professor, said the high court “has been hostile to administrative agencies, so it may not be the best thing for the FDA to lead the charge.”
Parsigian, the lawyer for GenBioPro, said he expected a prolonged legal battle. “I think we will win in some courts, lose in other courts, and eventually we will get the question to the Supreme Court.”
More to Read
Get the L.A. Times Politics newsletter
Deeply reported insights into legislation, politics and policy from Sacramento, Washington and beyond. In your inbox twice per week.
You may occasionally receive promotional content from the Los Angeles Times.