After Ohio passed abortion law, risk of complications nearly tripled
- Share via
Women seeking medical abortions in Ohio experienced a higher rate of complications after the state implemented a law that put new restrictions on doctors who performed the procedure, according to a study published Tuesday.
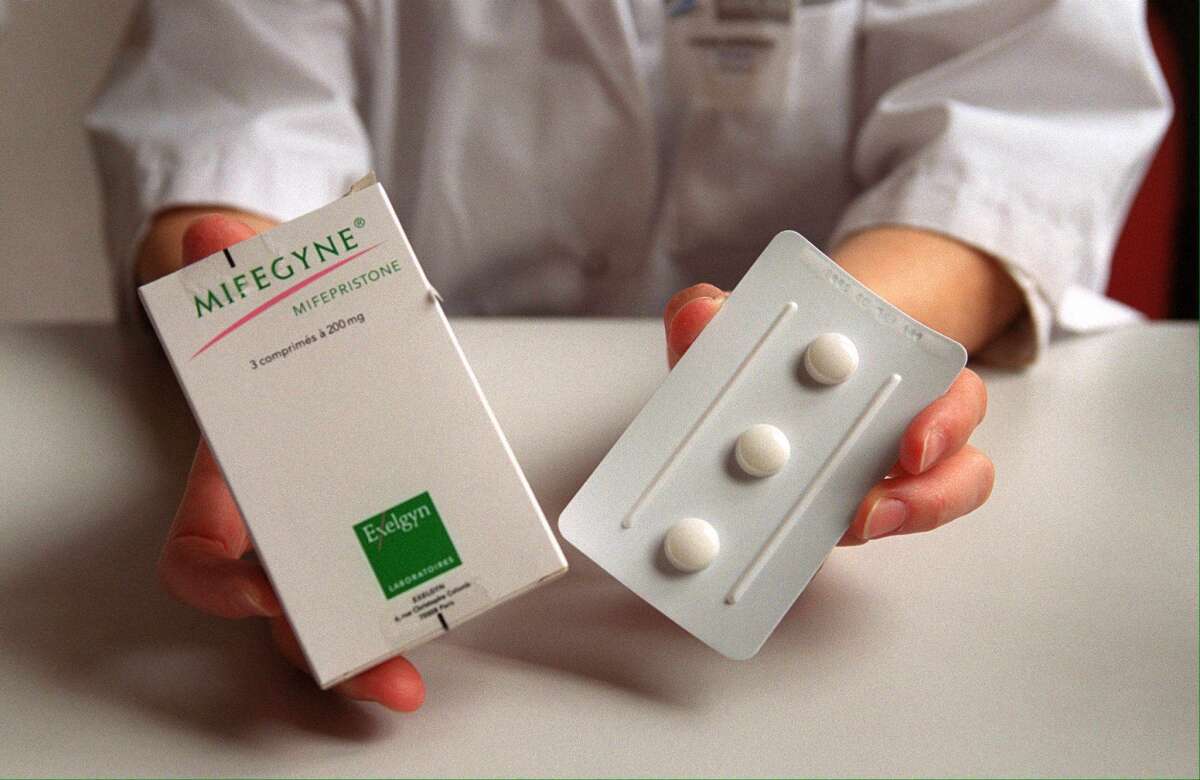
The law, which took effect in 2011, requires abortion providers to adhere to specific guidelines from the U.S. Food and Drug Administration when giving patients a combination of two drugs, mifepristone and misoprostol. The drugs have been shown to terminate early pregnancies safely and effectively.
The FDA protocol was set in 2000, with specific dosages of mifepristone and misoprostol. Within a few years, however, doctors realized that using a lower dose of mifepristone and a higher dose of misoprostol produced better outcomes for their patients.
The World Health Organization, the American Congress of Obstetricians and Gynecologists and the National Abortion Federation found shortcomings with the FDA’s protocol soon after it was issued, and began recommending changes as early as 2003. Healthcare providers throughout the U.S. followed their lead and prescribed the drugs according to the latest medical research rather than the dosages on the drug’s label.
The practice of prescribing drugs for use “off label” is both legal and commonplace. By one estimate, 21% of all U.S. prescriptions are intended for off-label use.
Before Ohio’s law went into effect, doctors in the state were able to decide what doses of the drugs were most effective based on the latest research and best practices. Afterward, doctors lost that flexibility and were forced to follow the FDA’s outdated protocol.
To see what effect that had on patients, researchers examined the medical records of 2,783 women who had medication abortions at one of four clinics in Ohio between 2010 and 2014.
The researchers found that women were nearly three times more likely to require additional intervention after the law was implemented than they were before.
Before 2011, doctors had to provide some kind of additional care in 4.9% of cases. Usually, this meant administering an additional dose of misoprostol or using suction to remove the fetus from the uterus. After the law was enforced, measures like these were required in 14.3% of cases, according to the study.
In addition, the incidence of side effects nearly doubled after the law took effect. When doctors were able to use their own judgment, 8.4% of women experienced problems like nausea or vomiting. After doctors were required to stick to the FDA’s regimen, 15.6% of women had such problems.
The average price patients paid for medication abortions went up as well, from $426 in 2010 to $551 in 2014, the researchers found. In part, that’s because the minimum number of required doctor visits rose from three to four.
“There is no evidence that the change in law led to improved abortion outcomes,” the study authors wrote in PLOS Medicine. “Indeed, our findings suggest the opposite.”
The FDA updated its guidelines in March, approving new labeling for Mifeprex (the brand name for mifepristone) that is more in line with current practice and makes the drug easier for women to use.
The new label extends the amount of time a pregnant woman can take the drug from seven weeks after her last menstrual period to 10 weeks. It also reduces the recommended dose from 600 milligrams to 200 milligrams, which reduces side effects and makes it less expensive.
Finally, the new labeling says women can take misoprostol (sold as Cytotec, among other brand names) at home rather than in the presence of a physician.
The FDA’s action means that women in Ohio seeking medication abortions can now receive care in line with the most up-to-date research. But that could change if the agency doesn’t keep up with evolving medical practices.
“This law will continue to require physicians to provide care that may fall below the accepted standard of care, placing them in an ethical dilemma,” they wrote.
North Dakota, Texas and Arizona have abortion laws similar to Ohio’s. State legislatures in Arkansas and Oklahoma passed similar laws, but those were blocked by a court order.
The study was led by Ushma Upadhyay, an associate professor in the Department of Obstetrics, Gynecology and Reproductive Sciences at UC San Francisco.
Do you love science? I do! Follow me @DeborahNetburn and “like” Los Angeles Times Science & Health on Facebook.
MORE IN SCIENCE
Pediatricians urge states to get tough on parents who don’t want to vaccinate their kids
3.2 million years after her death, autopsy reveals that ‘Lucy’ died of devastating fall from tree
The aging paradox: The older we get, the happier we are







