FDA clears device that lets paraplegics walk again
- Share via
A device designed by a quadriplegic Israeli entrepreneur to increase his own mobility, and in use already to get wounded U.S. military personnel up and walking again, has won clearance from the Food and Drug Administration for broad marketing in the United States.
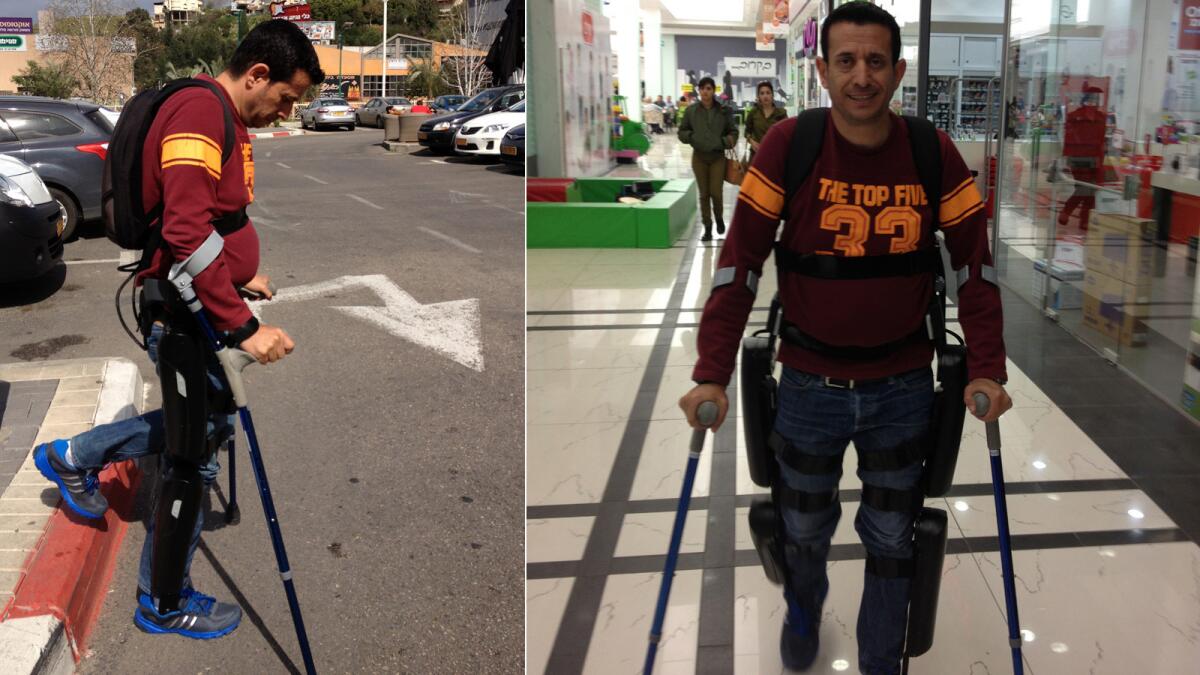
The ReWalk system is sometimes called an “exoskeleton” for those whose legs will no longer move at the command of signals from the brain. It consists of a fitted metal brace that supports the legs and part of the upper body. Its motors supply movement at the hips, knees, and ankles, and its computer and power supply are housed in a backpack worn by the user.
Using a wireless remote control worn on the wrist, the user of this “wearable device” can command the ReWalk system to aid him or her in standing up, sitting down or walking. Crutches provide the user with additional stability while walking, standing and rising from a chair.
A version of the system is widely used already in rehabilitation centers across the United States. But the ReWalk system cleared by the FDA on Thursday is for use by individuals outside of a rehab setting -- in their homes and communities. Its manufacturer said the system will be available immediately, with a price tag of $69,500.
The FDA’s approval specifies that the newly cleared ReWalk system is for use by paraplegics who have spinal cord injuries between the seventh thoracic vertebra and the fifth lumbar vertebra. When it is used in a rehabilitation setting, the agency said it may be used to assist people with injuries between the fourth and sixth thoracic vertebra.
ReWalk already is in wide commercial use in Europe and in Israel, where it was designed by Amit Goffer and is built by Argo Medical Technologies Inc., the research and development firm Goffer founded in 2001. Since then, Argo has expanded to open more than 50 ReWalk centers and train more than 400 ReWalkers around the world.
In 2012, Briton Claire Lomas became the first person to use the ReWalk to complete a marathon. And last fall, a ReWalk device arrived at Camp Pendleton in California for Capt. Derek Herrera, a Marine who was shot and paralyzed in 2012 while leading his special operations team on patrol in the Helmand River Valley in Afghanistan. Herrera, a staff officer who returned to work at 1st Marine Special Operations Battalion, demonstrated the use of the ReWalk system as an aid to both training and everyday work.
In addition, two wounded soldiers and a sailor were among the disabled patients whose experiences with ReWalk prompted the FDA’s clearance decision.
“Innovative devices such as ReWalk go a long way towards helping individuals with spinal cord injuries gain some mobility,” said the FDA’s Christy Foreman, director of the Office of Device Evaluation, on Thursday. “Along with physical therapy, training and assistance from a caregiver, these individuals may be able to use these devices to walk again in their homes and in their communities.”
Interested in human enhancement? Follow me @LATmelissahealy.






