Blood plasma appears safe for COVID-19 patients in early trial results
- Share via
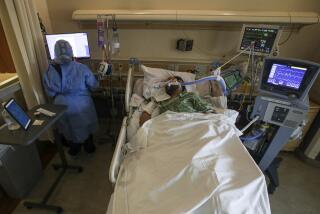
Taking antibody-rich blood plasma from people who have recovered from coronavirus infections and transferring it to patients who are severely ill with COVID-19 does not appear to increase their risk of dying from the disease, according to initial results of a nationwide clinical trial.
What’s more, the findings offer an early hint that the treatment may indeed save lives.
The early safety reading, posted this week to the MedRxiv website, likely clears the way for larger clinical trials to test two potential uses for plasma harvested from recovered COVID-19 patients: as a treatment for those in the midst of fighting a coronavirus infection, and as a way to protect essential workers who are routinely exposed to the virus from becoming seriously ill.
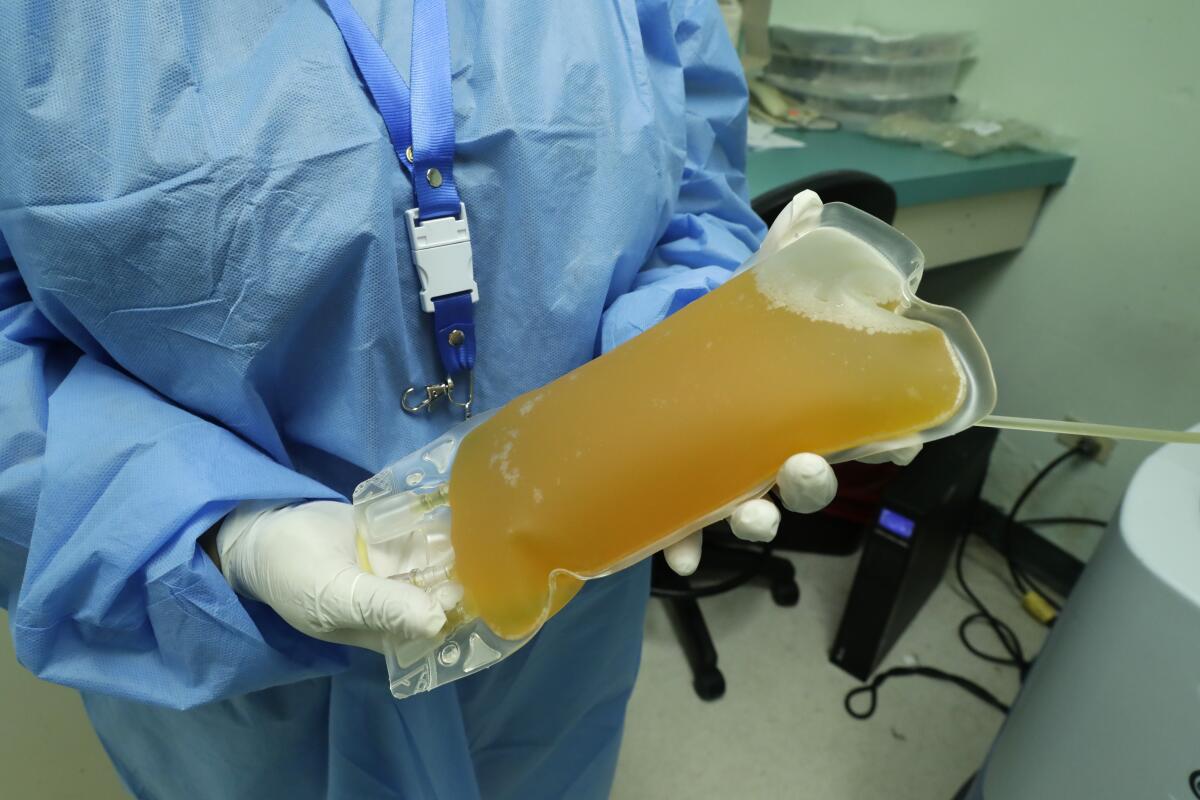
In the six months since a new coronavirus began sickening residents of Wuhan, China, scientists across the globe have sought to tap the power of recovered patients’ antibodies to treat the newly infected. For infected patients, they hope a transfusion of so-called convalescent plasma will speed recovery and increase the odds of survival.
In addition, the antibodies might give healthcare workers and first responders at least a temporary vaccine-like protection known as “passive immunity,” researchers have suggested.
But unlike a vaccine, which is not expected to become available before 2021, the supply of convalescent plasma is growing every day as more people join the ranks of the recovered. More than 1.5 million people worldwide have battled the coronavirus and survived, according to experts tracking the pandemic at Johns Hopkins University.
If further research bears out that it’s safe and effective, both treatment and prophylaxis with convalescent plasma could become common almost immediately since blood centers have already begun collecting blood from recovered individuals.
For now, the new results should be interpreted with caution because they have not yet been published in a peer-reviewed medical journal or thoroughly reviewed by independent scientists.
“It’s a long way away from showing that this thing is going to be a game changer,” said Dr. Michael Joyner of the Mayo Clinic, who is leading the trial.
“It’s a first step to prove that it’s safe, and it sets the stage for efficacy trials,” he added. “But we were very pleased.”
Scientists racing to stave off a tidal wave of coronavirus infections show renewed interest in a little-known medicine: convalescent plasma.
While still considered experimental for COVID-19, the approach has proved effective in battling infectious diseases for more than a century. Convalescent plasma has rescued patients afflicted with diphtheria, smallpox and polio, as well as more modern scourges like Ebola. But it remained unclear whether donors’ plasma could be safely infused into patients suffering from an infection that attacks the lungs, heart, kidneys and blood.
In the first of dozens of U.S. trials of convalescent plasma planned, researchers set out to ensure that the sickest COVID-19 patients who got the treatment were no more likely to die than similar patients who did not get it.
To date, roughly 11,000 Americans have received experimental transfusions of convalescent plasma under a special wavier from the Food and Drug Administration. The new findings are based on the first 5,000 of those patients, all of whom had severe or life-threatening COVID-19 or were at risk of developing it.
Joyner and his colleagues recorded and tallied all “adverse events” — everything from a case of hives to death — that followed a transfusion of between 200 and 400 milliliters of plasma.
In the four hours after receiving a transfusion, fewer than 1% suffered a serious adverse event, including decreased oxygenation, fluid buildup in the lungs or a severe allergic reaction. Any of those outcomes might be attributed to known complications of the plasma transfusion. But they could also be blamed on the natural progression of coronavirus infection, especially in patients who have become critically ill.
Overall, 15 of the 5,000 died within four hours of their transfusions. Three of those deaths were judged as possibly related to the transfusion, and a fourth was judged to be “probably” related.
An additional 21 severe adverse events that did not end in death were reported.
Stunning visualizations show what the novel coronavirus, SARS-CoV-2, looks like at an atomic level of detail.
The analysis was not intended to flesh out the treatment’s effectiveness, but the trial’s early signals gave researchers cause for “optimism,” they wrote. Seven days after these very sick COVID-19 patients received their transfusions of antibodies, 85% of them were still alive.
The trial, like many that are testing COVID-19 treatments, had no control group. Instead, the researchers — a group of doctors from the Mayo Clinic, Michigan State University and Johns Hopkins — compared their results with death rates seen in early studies of COVID-19 patients in China and New York.
Death rates in those studies ranged widely, both because the severity of patients’ illness was not uniform, and because patients were tracked for varying lengths of time before mortality rates were recorded.
In Wuhan, roughly 15% of all patients hospitalized in the pandemic’s early days succumbed to the disease.
Among the subset of patients classified as critically ill, death rates were much worse. In New York City, a study of COVID-19 patients found that 88% of 320 who required mechanical ventilation died, as did 61% of 52 critically-ill patients in Wuhan.
In the U.S. trial, all 5,000 patients had been hospitalized with COVID-19, and 81% had “severe or life-threatening” illnesses. The remaining 19% who got the experimental treatment were considered very likely to progress to that point.
Two-thirds of the treated patients had already been admitted to intensive care when they received the convalescent plasma transfusions, and 16.7%, of those patients died, the researchers reported. Among the patients who had not been admitted to the ICU, the death rate was 11.2%.
The death rate seen in treated patients was “not alarming,” the study authors wrote, especially considering the circumstances under which many of the experimental transfusions were performed. Some of the procedures “may be characterized as attempts at rescue or salvage therapy in patients admitted to the ICU with multi-organ failure, sepsis and significant comorbidities,” they wrote.
Joyner said there is reason to believe that less critically ill patients will tolerate the plasma infusions better, and that they may benefit more from its boost to the immune system.
As the new treatment approach moves to the next stage, several trials will gauge its effectiveness in newly-infected patients and those whose symptoms are more mild.
“That can only help” to clarify whether, and by how much, plasma from the recovered works to lessen the severity and duration of infection, as well as to save lives, Joyner said.