Two women win Nobel Prize in chemistry for work on CRISPR gene-editing system

- Share via
Two women from different continents who met at a cafe in Puerto Rico were awarded the Nobel Prize in chemistry Wednesday for their pioneering work on CRISPR, a gene-editing tool that gives scientists the power to change the behavior of living cells and potentially cure diseases.
UC Berkeley biochemist Jennifer A. Doudna and Emmanuelle Charpentier, director of the Max Planck Unit for the Science of Pathogens in Berlin, harnessed an ancient immune response found in bacteria and other single-celled organisms and shaped it into an elegant pair of “molecular scissors,” the Nobel Committee said. The tool has transformed the life sciences in just eight years.
With CRISPR, researchers can alter specific sequences on the human genome with the hope of priming patients to fight cancer or curing diseases such as sickle-cell anemia. It also paves the way for improving plants and livestock by imbuing them with greater disease resistance, and for safer transplants of animal organs into humans.
A researcher in China said he used CRISPR to tweak the DNA of human embryos so they would grow up to have immunity to HIV. The controversial work produced three babies and resulted in a three-year prison sentence for the scientist.
“There is enormous power in this genetic tool, which affects us all,” said Claes Gustafsson, chair of the Nobel Committee for Chemistry. “It has not only revolutionized basic science but also resulted in innovative crops and will lead to groundbreaking new medical treatments.”
He added that “the enormous power of this technology means that we need to use it with great care. But it’s equally clear that this is a technology and method that will provide humankind with great opportunities.”
CRISPR is having a moment.
Charpentier, who was born in France, said she “was very emotional” after learning that she and Doudna had won.
It is the first time the prize has been shared by two women. Charpentier said she hoped it would send “a positive message” to girls and young women “who would like to follow the path of science and ... show them that, in principle, women in science can also be awarded prizes but, more importantly, that women in science can also have an impact through the research.”
Doudna said she realized early on that the two of them were on to something: “You never know where science is heading, but I kind of had a feeling it would be a big deal.”
She was right.
“The seminal paper they published together has been cited more than 9,500 times — approximately once every eight hours since its publication in 2012,” said David Liu, a chemical biologist at Harvard University who has worked to perfect the system.
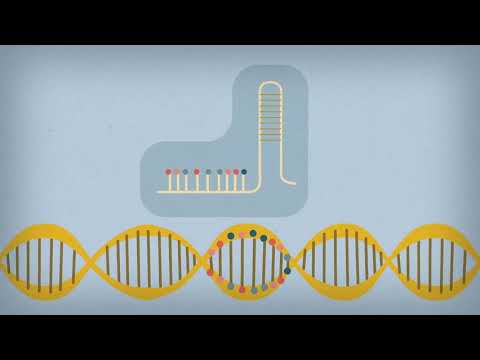
CRISPR provides a way to find and alter specific DNA sequences.
CRISPR is made up of two basic components. The first is a piece of RNA that locates a predetermined sequence of DNA in an organism’s genome that scientists want to alter. The second is a type of protein called an enzyme that attaches itself to the target section of DNA and splices it.
When a DNA strand has been cut, the cell immediately sets to work to repair it. Scientists can exploit this impulse by adding another piece of DNA to the CRISPR complex that the cell can use to mend the break. This makes it possible to insert new genes into a DNA strand.
CRISPR can also be used to disable faulty genes and even change a single letter on a strand of DNA.
The system takes advantage of the way bacteria find and destroy invading viruses by storing sequences of viral DNA within their own DNA. Those sequences are bookended by a repeating sequence of letters that give CRISPR its name — Clustered Regularly Interspaced Short Palindromic Repeats. (The first CRISPR systems discovered were indeed partly palindromic; however, scientists later found that this is not universally true.)
Doudna and Charpentier, who met in 2009 while attending a scientific conference, were not the first to spot these odd stretches of DNA, nor were they first to realize that it was part of a bacterial immune system. Indeed, by the time they published their revolutionary paper, other scientists had shown that the system required a piece of RNA and an enzyme.
But the two women were the first to identify that another piece of RNA was required to bind the two together, and found a way to streamline them. With all the components in place, the system could be programmed to find and cut any piece of DNA a scientist desired, making it a more efficient genome editor than had ever been seen before.
Talk about precision gene editing.
Within six months, six independent research teams had demonstrated that the CRISPR gene editing system would work in animal cells.
Most early CRISPR systems used an enzyme called Cas9 to make the cut. More recently, scientists have been experimenting with other enzymes as well.
“My greatest hope is that it’s used for good, to uncover new mysteries in biology and to benefit humankind,” Doudna said.
Last year saw the first attempt in the U.S. to use the CRISPR tool to treat cancer patients. Doctors took immune-system cells from the patients’ blood, altered the DNA inside those cells to help them recognize and fight cancer, and reintroduced them into the patients. Whether the treatment will improve their chances of survival is still too early to say.
The ability to edit genes is fraught with difficult ethical questions, which Doudna examined in her book “A Crack in Creation: Gene Editing and the Unthinkable Power to Control Evolution.” The book, which she wrote with her former student Samuel H. Sternberg, said that CRISPR technology offered “both the greatest promise and, arguably, the greatest peril for the future of humanity.”
“It’s widely available and it’s accessible to people around the globe,” Doudna told the Los Angeles Times when the book was published in 2017. “That’s what’s wonderful about it, but also one of the real challenges. How do you control something like that?”
A day after a blockbuster report that researchers had edited harmful genetic mutations out of human embryos in an Oregon lab, an international group of genetics experts urged scientists against taking the next step.
The CRISPR breakthrough has spawned a protracted legal fight with the Broad Institute at MIT over patents for the technology. Other scientists — particularly Feng Zhang of the Broad — have conducted significant research on the technology, but the U.S. patent office recognized Doudna as the inventor last year and awarded several key patents to UC Berkeley.
Doudna has helped found a number of CRISPR-based companies, including Mammoth Biosciences, Caribou Biosciences, Intellia Therapeutics and Scribe Therapeutics. Charpentier is co-founder of CRISPR Therapeutics.
“There comes a point when discoveries need to be translated in a way that is difficult or impossible to do in an academic setting,” Doudna said. “It is really exciting to see these companies develop real solutions in biomedicine and other areas using CRISPR technology.”
The Nobel Prize in chemistry frequently honors research that leads to practical applications in wide use. Last year’s winning trio pioneered the lithium-ion battery, a lightweight, rechargeable power supply that is ubiquitous today.
The chemistry award is one of the six 2020 Nobel Prizes that are being announced through Monday. Andrea Ghez, an astronomer at UCLA, and two others won the physics prize Tuesday for their work on black holes, including the supermassive black hole at the center of the Milky Way galaxy. The prize for medicine was given Monday to scientists who discovered the hepatitis C virus.
The other Nobel Prizes are awarded in literature, peace and economics.
The award comes with a gold medal and more than $1.1 million, drawn from a bequest left by Swedish inventor Alfred Nobel, who established the prizes that bear his name.








