Moderna to expand size of COVID-19 vaccine study in younger children to better detect rare side effects
- Share via
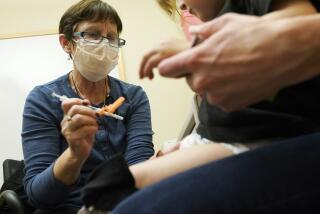
WASHINGTON — Moderna said Monday that it plans to expand the size of its COVID-19 vaccine study in younger children to better detect rare side effects, such as a type of heart inflammation recently flagged by U.S. health authorities.
The company said it is in talks with the Food and Drug Administration to enroll more study participants younger than 12. It had intended to test the vaccine in about 7,000 children, with some as young as 6 months. The Cambridge, Mass.-based company said via email it hasn’t decided how many children might be added.
The announcement comes as U.S. COVID-19 cases are rising and schools prepare to welcome students back to classrooms. At the same time, regulators continue to review cases of a rare type of heart inflammation called myocarditis that has been reported in a small number of teenagers who got the Moderna or Pfizer shots.
Moderna says its COVID-19 vaccine strongly protects children as young as 12, and will submit trial data to U.S. regulators next month.
Pfizer said Monday that if it makes changes to its vaccine testing in children, it will provide an update then. The New York-based company is testing its vaccine in as many as 4,500 children in the United States and Europe.
The FDA said in a statement it could not comment on its discussions with companies, but added, “we do generally work with sponsors to ensure the number of participants in clinical trials are of adequate size to detect safety signals.”
The news was first reported by the New York Times.
U.S. officials and independent medical experts said last month the benefits of the vaccines far outweigh the risks of the side effect, which has been reported in several hundred people younger than 30. But any vaccine safety issues could slow uptake, particularly among parents wary of taking any health risk with their children.
Currently, Pfizer has the only U.S. vaccine authorized for children 12 years and older, while Moderna is expecting an FDA ruling on its application in the coming days.
Teens receive the same dose as adults, but younger children may need smaller doses. That additional complexity adds time to drugmakers’ research and application timelines.
The recommendation means many middle schoolers and virtually all high school students have the chance to be vaccinated before the next school year.
Moderna said Monday that it expects to have enough data to apply for FDA authorization in younger children by late this year or early 2022.
Pfizer has previously said it expects to apply in September for children ages 5 through 11. Results for two younger age groups that began testing a little later should be available by October or November, according to the company.
More to Read
Sign up for Essential California
The most important California stories and recommendations in your inbox every morning.
You may occasionally receive promotional content from the Los Angeles Times.