FDA Approves Use of Abortion Pill
- Share via
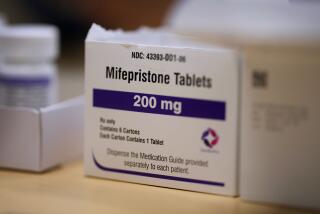
WASHINGTON — The Food and Drug Administration gave final approval Thursday to the abortion pill RU-486, offering women the option of terminating early-stage pregnancies with drugs instead of surgery and easing the way for more doctors to provide abortion services.
The drug, approved for use through the first seven weeks of pregnancy, will be marketed as Mifeprex and “the early option pill.” Already available in much of Western Europe, it could reach U.S. doctors in about a month.
The pill is part of a two-drug regimen that works by causing a miscarriage and is the first approved alternative to surgical abortions, which in most cases rely on suction equipment to end a pregnancy. The drug has the potential to make abortions more private, moving them from the clinics that are frequent targets of protests and into the offices of thousands of gynecologists and family doctors.
Abortion rights supporters, who worked for 12 years to bring the drug to U.S. consumers, called the FDA decision a milestone in women’s health, nearly as significant as the arrival of the birth control pill 40 years ago. Currently, 86% of U.S. counties don’t have an abortion provider, but that could change under FDA rules that allow nearly any doctor to offer Mifeprex.
The drug also could encourage women to have earlier abortions, its advocates said. Thanks to increasingly sensitive pregnancy tests available in drugstores, women are finding out earlier than ever that they may be pregnant. Now, many of them will be able to obtain an abortion, without invasive equipment or anesthetic, soon after getting the news.
The FDA announcement immediately became an issue in the presidential campaign, with anti-abortion groups accusing the Clinton administration of trying to marshal its allies in the abortion rights movement. They called attention to the drug’s side effects, which include bleeding and cramping, and said that changing the method of abortion does not make the procedure any less immoral.
“We will not tolerate the FDA’s decision. . . . We will shout it from the rooftops: RU-486 kills innocent human persons,” said Judie Brown of the American Life League, an anti-abortion group. “RU-486 is a chemical assault weapon aimed at the tiniest babies.”
President Clinton said the FDA had focused only on patient safety. He declined to portray the decision as a personal victory, though he was instrumental in initiating the FDA review. On his third day in office eight years ago, Clinton ordered federal health officials to find ways to bring the drug to U.S. consumers, in essence reversing the policy of his predecessor, George Bush.
Bush Fears Increase; Gore Praises Choice
“The decision to be made under our law is whether the drug should be approved by the FDA on grounds of safety . . . ,” the president told reporters Thursday. “I think it ought to be treated as the scientific and medical decision it was, and we should respect the fact that it was a nonpolitical inquiry.”
Texas Gov. George W. Bush, the Republican presidential nominee and son of the former president, said the FDA decision is “wrong.” But he did not say what he might do as president to try to restrict the drug or remove it from the market.
“I fear that making this abortion pill widespread will make abortions more and more common, rather than more and more rare,” Bush said in a statement. “As president, I will work to build a culture that respects life.”
Vice President Al Gore, the Democratic presidential nominee, who favors abortion rights, said he was “pleased” by the FDA announcement. “Today’s decision is not about politics but the health and safety of American women and a woman’s fundamental right to choose,” Gore said in a statement.
Mifeprex already has been approved in 18 countries, and more than 500,000 women have used it in Europe alone. In France, the first country to approve the drug more than a decade ago, it accounts for 70% of early-stage abortions and one-third of all abortions. But it has not increased the total number of abortions there, U.S. sponsors of the drug said.
Based on France’s experience, U.S. advocates said, the drug will encourage women to have earlier abortions but not more abortions.
The drug’s path to approval in the United States had been hampered by opposition from anti-abortion groups. In addition, investors and manufacturers were reluctant to risk lawsuits and abortion protests for a drug with likely revenues of tens of millions of dollars a year, not the billions of dollars that pharmaceutical makers often seek.
In fact, fear of anti-abortion protests prompted unusual measures by the FDA and Danco Laboratories LLC, the small, privately held New York firm that is marketing Mifeprex. The FDA took the uncommon step of declining to release the names of its medical officers who reviewed the drug. A Danco news conference in New York City featured tight security Thursday, which included police guards.
Danco has declined to reveal who has financed the company and where the drug will be manufactured, a stance that a spokeswoman said is standard in the pharmaceutical industry but which also may insulate the company from protesters. Moreover, the company will distribute the drug to patients directly through doctors, not through pharmacists. The company said this is the most effective way to ensure that the drug is used properly. But that also makes it harder for protesters to disrupt the supply chain. Nor did Danco say how much the drug will cost.
Doctors Still Must Follow State Laws
While the drug could prompt more doctors to offer abortion services, abortion rights groups said that shift will come gradually. At least 14 states regulate abortion clinics, in some cases with special licensing requirements and rules on air flow and doorway width. Doctors will have to determine whether those laws apply if they offer only drug-induced abortions. In addition, doctors will have to comply with state laws on parental consent and notification for minors. And they may not want to take on the added responsibility of offering the drug--or the risk of drawing protesters.
“This is not a magic pill, and it’s not going to end the abortion debate in the United States,” said Vicki Saporta, executive director of the National Abortion Federation, a league of abortion providers. “It does shift the debate to very early abortions, where there is a great deal of public support. And so I think that [abortion opponents] will now have a more difficult time trying to rally public opinion behind them.”
One anti-abortion group said that there would be no slowdown in its campaign of picketing doctors who provide abortions, no matter what procedure they use. “The doctor-turned-abortionist is an accomplice to murder, and we’ll treat them as murderers,” said Troy Newman, director of Operation Rescue West, which targets abortion providers in 11 Western states, including California. “We’ll treat them as a dirty abortionist. . . . I will continue to stigmatize doctors, OB/GYNs, that continue to practice murder.”
Mifeprex, also known by the scientific name mifepristone, in essence causes a pregnancy to miscarry by depriving the uterus of a natural hormone needed to sustain an embryo. It is used with a second drug, misoprostol, which is taken two days after Mifeprex and causes the uterus to contract and expel the embryo. Under the FDA-approved treatment plan, women who want to use it will have to make three trips to a doctor’s office over a two-week period.
The FDA has approved Mifeprex for use during the first seven weeks of pregnancy, a period when one-third of U.S. abortions are conducted. However, doctors can legally offer it as they wish. Some have already reported that it can be highly effective through the first nine weeks of pregnancy, when more than half of U.S. abortions occur.
Concerns Over Who Will Produce Drug
In approving the drug, the FDA chose not to adopt several restrictions that it had mentioned privately this summer to Danco Laboratories. Among them were requirements that doctors be trained in traditional abortions and have admitting privileges at nearby hospitals before offering the drug.
Critics said this would have sharply curtailed access to the new pill, especially in rural areas. On Thursday, the FDA said physicians could offer the drug if they are able to accurately determine how long a woman has been pregnant and have made plans for providing surgical care in case of emergencies.
Opponents of the pill have pursued several courses of action in Congress, with little success. Attempts to restrict the FDA from spending money to review RU-486 have failed.
Abortion opponents are focusing on reports Danco is using a Chinese manufacturer to produce the drug.
Citing those reports, which first appeared in the Wall Street Journal and have not been confirmed by Danco, Sen. Jesse Helms (R-N.C.) recently asked the Department of Health and Human Services to answer a variety of questions about the manufacturer. Among them: whether it is owned by the Chinese government, uses coercive or slave labor, or produces drugs used in the Chinese family-planning program.
“Who is exporting this pill and what it actually contains is certainly one area Congress will look into,” said Rep. J.C. Watts Jr. (R-Okla.), the House Republican Conference chairman.
The drug was created in the early 1980s by the French firm Groupe Roussel Uclaf--representing the RU in the drug’s laboratory name. French bishops and the Vatican urged Roussel Uclaf to abandon the drug, and it came to market in France in 1988 only under pressure from the French government. A boycott threat and other factors made Roussel Uclaf reluctant to bring it to the U.S. market.
With the encouragement of the Clinton administration, Roussel Uclaf in 1994 donated U.S. rights to the drug to the Population Council, a New York nonprofit group. The council, in turn, licensed Danco to bring it to the U.S. market.
*
John-Thor Dahlburg in Paris and John J. Goldman in New York contributed to this story.
(BEGIN TEXT OF INFOBOX / INFOGRAPHIC)
How Abortion Pills Work
The FDA will allow mifepristone to be distributed only to doctors trained to accurately diagnose the duration of pregnancy and to detect problem pregnancies. Physicians must also ensure women have access to surgical abortions in rare cases where they are needed. Here’s how the pills work:
Treatment Method
Obtain first dose from doctor, then return two days later for second drug. Twelve days later, patient must be examined to ensure abortion is complete to prevent infection.
Possible Side Effects
Bleeding and cramping are common; nausea, diarrhea and headache are less common. Bleeding and spotting last an average of nine to 16 days and may continue for 30 days.
More to Read
Sign up for Essential California
The most important California stories and recommendations in your inbox every morning.
You may occasionally receive promotional content from the Los Angeles Times.