Column: U.S. prosecutors sue to shut down clinics selling unproven stem cell treatments in California and Florida
- Share via
The U.S. Department of Justice filed lawsuits Wednesday aimed at shutting down clinics in Florida and California that have marketed unapproved and unproven “stem cell” treatments to thousands of patients, some of whom suffered severe adverse reactions.
The lawsuits are the most aggressive steps yet taken by government regulators against rapidly proliferating stem-cell treatment clinics pitching scientifically unproven therapies to desperate patients, some with terminal illnesses. The lawsuits were filed at the request of the Food and Drug Administration, which last year announced that it would take a stern approach to what it described as the manufacture and use of unlicensed drugs.
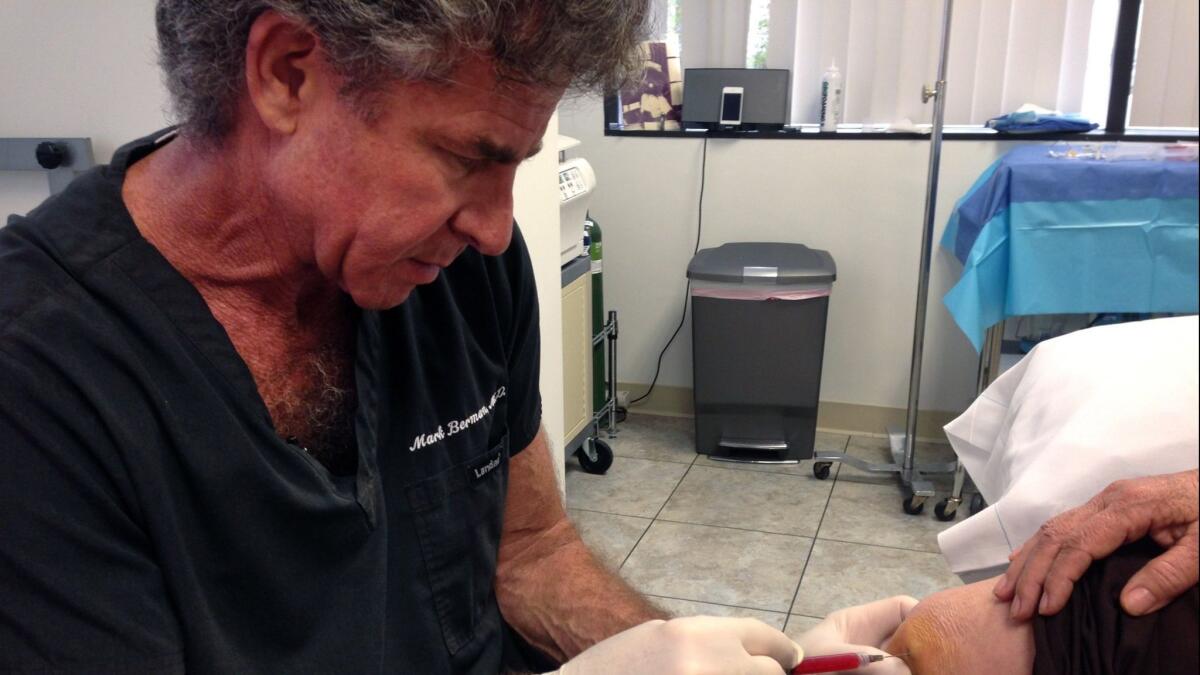
The targets of the California lawsuit are California Stem Cell Treatment Center and its proprietors, physicians Mark Berman, a cosmetic surgeon, and Elliott Lander, a surgeon and urologist. The center has two locations, in Rancho Mirage and Beverly Hills. According to the complaint filed in federal court in Los Angeles, the center charges many patients thousands of dollars per treatment.
Marketing unproven and potentially unsafe treatments puts consumers at risk.
— Assistant U.S. Atty. Gen. Chad Readler
Lander and Berman have about 100 affiliated licensees around the country, which have treated more than 6,000 patients, according to the lawsuit. None of the treatments has been approved by the FDA, the lawsuit says, even though under the law they are considered drugs subject to FDA jurisdiction. Several patients suffered severe side effects from these treatments, the lawsuit alleges, including loss of consciousness and infections.
California Stem Cell Treatment Center was the target of an FDA action in August, when the agency said it seized nearly 500 doses of a smallpox vaccine that was to be combined with stem cells derived from body fat and administered to patients at the Beverly Hills and Rancho Mirage clinics.
The Florida lawsuit, filed in federal court in Miami, names U.S. Stem Cell Clinic and company officers Kristin Comella and Theodore Gradel. We reported on the clinic and Comella last month, raising questions about its treatments and Comella’s background, which includes a doctorate issued by an online “virtual” university in the Republic of Panama.
As many as four patients the Florida clinic treated for macular degeneration with stem cell injections into their eyeballs have suffered severe effects, including blindness. The clinic was the target of an FDA warning in August, after agency inspectors cited it for 14 “significant objectionable conditions” at the clinic, including failure to perform adequate testing to ensure that the substances it was injecting into patients were free from contamination.
The targeted clinics in both states purport to treat a wide variety of ailments by extracting stem cells from fat removed from the patients by liposuction. The cells are then reinjected in the patients, often the same day.
Lander and Berman say their procedure can treat “cancer, pulmonary disease, arthritis, stroke, ALS, and multiple sclerosis,” the Justice Department says. Comella’s clinic has claimed to treat “Parkinson’s disease, spinal cord injuries, stroke, pulmonary disease, and traumatic brain injury.”
None of these treatments has been validated through scientific or medical research. “Marketing unproven and potentially unsafe treatments puts consumers at risk,” Acting Assistant Atty. Gen. Chad A. Readler said in a statement.
Neither Berman nor Lander could be reached for comment Wednesday. Berman, however, asserted in an email after our column on Comella that “the FDA is trying to turn your personal cells into a pharmaceutically produced drug.”
Comella said in a statement Wednesday, “I remain steadfast that no government agency should deprive individuals of their right to harness the cells that exist in their body.”
Keep up to date with Michael Hiltzik. Follow @hiltzikm on Twitter, see his Facebook page, or email michael.hiltzik@latimes.com.
Return to Michael Hiltzik’s blog.
More to Read
Sign up for Essential California
The most important California stories and recommendations in your inbox every morning.
You may occasionally receive promotional content from the Los Angeles Times.