Here’s what coronavirus testing for a very sick patient looks like, from swab to result

- Share via
The patient was at risk.
A fever somewhere above 101. Hypoxia, shortness of breath. And two critical check marks for risk: advanced age and a compromised immune system.
For the record:
4:12 p.m. April 7, 2020A photo caption with a previous version of this article identified Mikkiel Cheng as a UCLA clinical lab technician. Cheng’s title is clinical lab scientist.
In the eyes of the medical staff, the cause could be bacterial, but odds were that a virus had found its way to the most receptive cells and hijacked their RNA. One became two, two became thousands, filling the lungs.
Another ILI, an influenza-like illness.
The patient, who had just arrived in the emergency department of the Ronald Reagan UCLA Medical Center in Westwood, had to be treated.
But treatment required precaution, and precaution required testing.
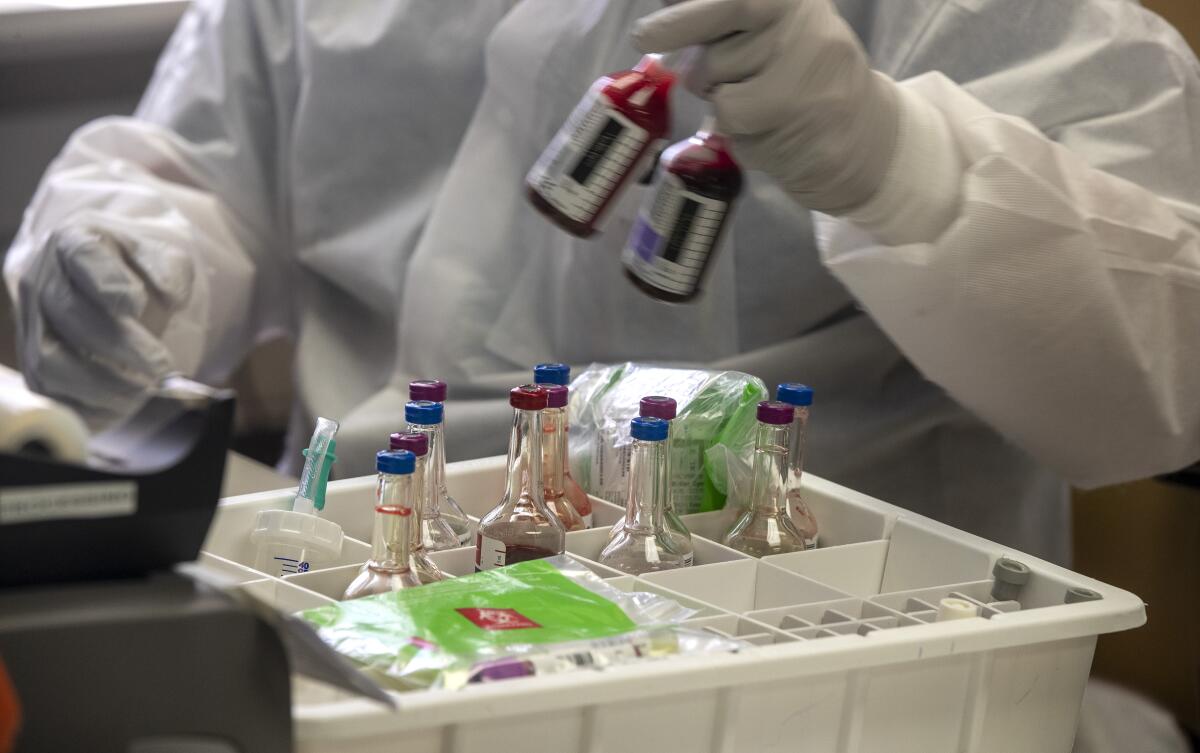
Three months since the novel coronavirus and COVID-19 appeared on the world’s stage, few modern illnesses have proved to be as contagious.
Since the first case — a viral pneumonia reported in China on Dec. 8 — was reported, the World Health Organization has recorded nearly 575,000 cases and nearly 26,500 deaths in 201 countries.
For an illness with no vaccine and limited treatment, testing and isolation are the best methods for stopping the spread, and even though the virus’ genome was sequenced within a month of that first case, testing in the United States has lagged, hindered by cumbersome bureaucracies, shortages and antiquated practices.
For Omai Garner, who directs clinical microbiology testing for UCLA Health, laboratories like his are central for ending this pandemic. Their protocol, initially developed in the 1980s, has been on the front line of viral diagnostic testing for almost two decades.
Of the viruses that Garner has encountered, the coronavirus is one of the most sinister, a Trojan horse disguising itself in symptoms of the flu.
The next few weeks are critical, he said, to get an accurate picture of where we are in this outbreak: “What you will hear from most healthcare professionals is we have no idea.”
He is skeptical of a new test, fast-tracked by the U.S. Food and Drug Administration and coming on the market next week, that promises greater convenience and faster results. Neither he nor his colleagues, he said, have been able to compare its results with their own.

“We need to do a lot more inquiry before we know how effective it will be,” he said, “because it is not based on the established standard for infectious disease diagnoses.”
In a series of phone interviews last week, Garner shared details about the testing protocol that his laboratory followed in its analysis of one possible COVID-19 case.
::
The emergency room moved quickly.
The mood that afternoon — just days after the city and county began shutting down nonessential activities — was calm yet wary. The number of walk-ins had dropped; the ambulance bay was quiet. An eerie sense of idleness had set in.
Treating this patient was a trained reflex of isolating, monitoring, administering sodium chloride and ordering battery of exams.
But the medical staff couldn’t help but wonder what lay ahead of them. They knew about Italy, the triage in those hospitals, and they were hearing of a flood of patients swamping hospitals in New York.
The door to the treatment room slid open and closed. Distant voices grew loud, then soft again.
A nurse in blue scrubs, face shield, mask and gloves held a plastic bag. With an easy tug, the seam tore open.
A swab and test tube filled with a pink-tinged solution were placed on a sterile tray. The same collection kit is used for the scores of pathogens — influenza, gonorrhea, whooping cough — that need identification for treatment.
The patient lay alone; new policy prohibited visitors.
The nurse explained the procedure, then guided the bristly swab — a lance-like Q-tip — through a nostril, farther and farther, until it reached the back of the throat near the adenoids where viruses often take hold amid the warm and moist tissue.
Pain shot through the patient, trying to breathe through a gauze of congestion. Eyes welled with tears.
The nurse extracted the swab — covered with a film of mucus, rich with the cells of the patient and a possible virus — and placed it into the accompanying test tube.
The gooey extraction loosened amid a solution of salt, carbohydrates and antibiotics designed to preserve the viability of the virus for the hours ahead.
A white label covered the tube, identifying the specimen with a series of numbers beginning with 20SA-83.
Placed in a biohazard bag, it flew by pneumatic tube to the basement, where it was placed in cooler bag, stored at 40 degrees to deter bacteria and fungus from growing.
Garner’s laboratory was a mile away.
::
Garner, 41, always knew he would be a virologist. Growing up near Milwaukee, he was drawn to the worlds under a microscope, but not until the 1995 movie “Outbreak” did he see his future.
“To a young African American teenager, the idea that Cuba Gooding Jr. was in this movie playing a hero in a suit, studying the Ebola and fighting it, well, I was hooked,” he said. “I wanted to be the one fighting the apocalypse.”
Years later, his understanding of viruses grew more nuanced. Less a scourge of humanity, they had a simple beauty: adaptable to their environment, flexible in their evolution and vital to the human species.
“If you look at the human genome, large segments of our DNA come from viruses,” he said. “We are human because of our interactions with viruses.”
That afternoon, Garner stopped by the lab. Long hours during the week — typically 7 a.m. to 5 p.m. — meant shorter hours on the weekend.

Standing in his office on the fourth floor before heading downstairs to the lab, he glanced out the window toward the Getty Center and the Santa Monica Mountains. Here we are in the midst of an unprecedented crisis, a threat to so many people, he thought, the look of the sky doesn’t change.
Outside, a van drove up the driveway to the lab’s second floor receiving area. The building on San Vicente Boulevard in Brentwood is undistinguished. In better times, a restaurant on the ground level serves Italian.
The van’s rear doors swung open. A medical courier carried four soft cooler bags containing 200 specimens into the lab.
Just three months ago, Garner first heard of a “mystery virus,” an “unknown infection,” spreading in China, but he didn’t give it a second thought. Viral outbreaks from unidentified pathogens happen all the time; a new one didn’t raise any immediate alarms.
At the time he was focused more on his wife and their first child, born Jan. 5.
But a week later his phone rang. A lab colleague, Shangxin Yang, was concerned. He knew people near the city of Wuhan.
“This is going to be bad,” Yang said.
::
Garner marked the date — Jan. 15 — when he first heard the phrase “novel coronavirus” and put two assistant directors and his senior specialists to work.
The UCLA Medical Center had long been the first stop for sick travelers arriving at Los Angeles International Airport, and the airport at the time was one of three in the country screening for the virus.
He wanted to develop testing in his laboratory and not depend on others. He had the machinery, mothballed for research, that he could dust off, and he worried that the U.S. Centers for Disease Control and Prevention would not be able to accommodate the number of cases that he expected to see.
The CDC was requiring collection kits be tested in Atlanta, a slow and cumbersome process. Then there was a shortage of collection kits, and when the FDA finally allowed other laboratories to do their own testing, only a few had the compatible equipment (and most of that was manufactured in Germany and China).
Garner was also concerned that the CDC criteria for screening were too narrow. “South Korea was having active cases,” he said, “but if someone traveled from South Korea and had the right symptoms, they didn’t get tested.”
The clincher, though, was the test itself.
“The CDC designed a beautiful test,” Garner said, “but it was a little over-elegant.”
Its flaw, he explained, was in requiring the presence of three viral genomic markers for a positive result. When health agencies around the country began running trials in late January, they found only two of the markers.
This defect led the CDC to revise and eventually drop one of the markers. Two would be enough, but by then it was Feb. 28. Critical time had been lost.
“When trying to get ahead of the curve of the outbreak, we were already well behind,” Garner said. “It is unfortunate what happened. We lost six to eight weeks for widespread testing.”
::
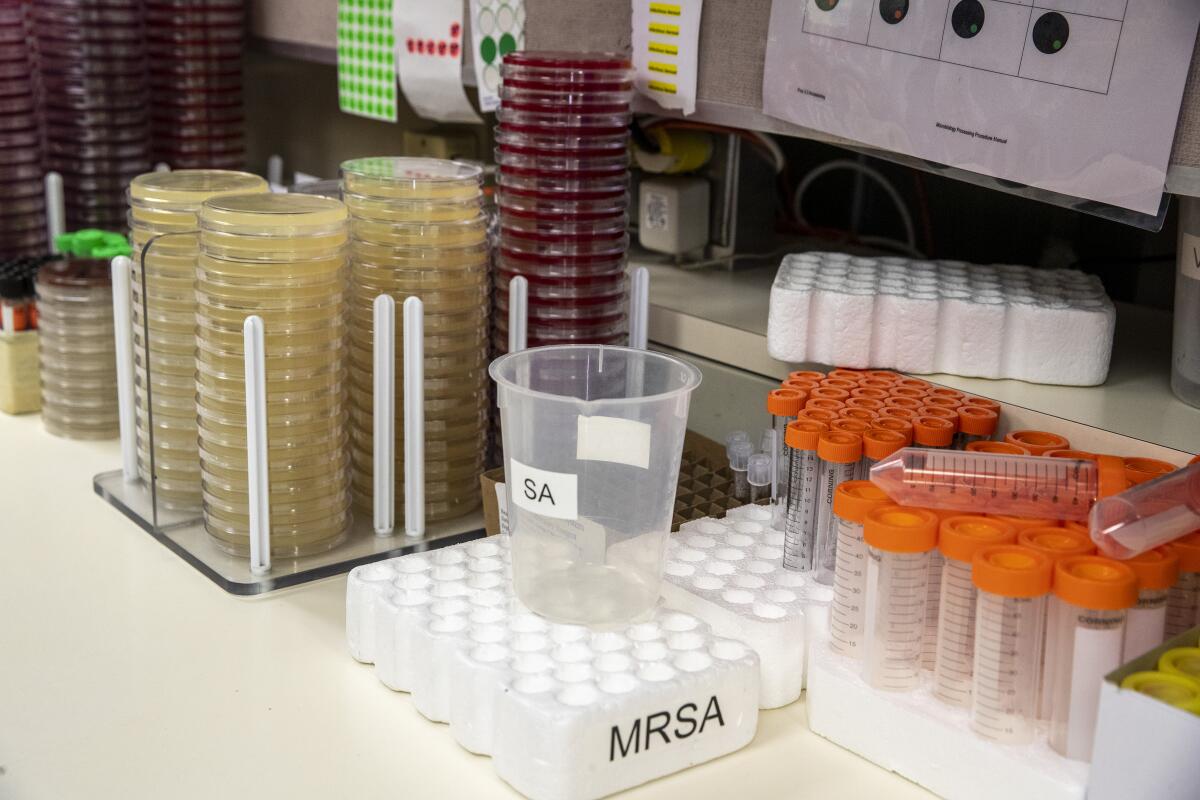
Within two hours of the patient arriving at the emergency room, the specimen inside No. 20SA-83 had reached Garner’s lab, and Carlos Azucena, a clinical laboratory scientist, started the protocol.
He guided his gloved hands under the glass partition of a bio-safety cabinet, slipping through a soft seal of ventilated air designed to keep pathogens from escaping into the room. Working slowly, he made sure the air barrier remained intact.
He placed No. 20SA-83 on a vortex spinner. The pinkish solution whirled and churned. He transferred it from the test tube into a small vial and lowered it into a Styrofoam rack. He soon had eight prepared specimens.
A small black disc, no larger than a CD, lay in front of him. It had eight pie-shaped compartments and looked like a fortune wheel. He opened a flap over one of the compartments, revealing two shallow wells.
Using the calibrated pipette, he drew out 50 microliters — just a drop — from No. 20SA-83 and released it into one well.
He turned to another set of vials, containing a reagent, the enzymes and chemical markers specially designed to reveal the presence of the coronavirus. He drew out a drop and placed it in the other well.
After filling the eight compartments and closing their flaps, he removed the disc from the cabinet and inserted it into a thermocycler, a hooded gray machine just slightly larger than a pressure cooker.
Azucena closed the lid, and the disc began spinning, reaching nearly 1,600 rpm. The temperature inside rose to more than 200 degrees Fahrenheit.
For 90 minutes, these two extremes pitted specimen with reagent in a modulated sequence known as a polymerase chain reaction. The film of mucus, once covering the tip of the swab and containing the patient’s cells, began to break apart.
Their RNA converted into a single strand of DNA, awash with the enzymes and chemical markers that would target the coronavirus, and, if present, emit an impulse of light as it bonded, a brief and measurable florescence.
“In its beauty, it is hard to understand the impact of the PCR [polymerase chain reaction],” Garner said. “It is like the Rosetta stone, showing us how to read the information inside a cell.”
As the thermocycler completed the test, Azucena reviewed the results.
::
The next day at 7 a.m., Garner opened the daily report from the laboratory. In the previous 24 hours, his team had conducted 150 tests; a week before, just 30 tests. The increase was unprecedented, but he trusted the methodology.
The company that formulated the reagent was headquartered near Milan, and the outbreak in northern Italy guaranteed experience with the virus.
His eyes paused on No. 20SA-83.
Another positive.
By then, he guessed, the patient was in isolation in the hospital. The attending physician, who received this news within minutes of Azucena’s review, could have begun antiviral treatments.
The hospital could screen its healthcare workers and provide its infectious disease specialists with another data point in charting the progression of the virus in the community.
Since ramping up testing, his laboratory had found that 10% of the tests among people with moderate to severe illness were positive. The rise or fall of this number is the best indication whether social isolation is slowing the spread.
“This is the canary in the coal mine for how bad things are getting,” he said.
He only wishes more laboratories had this capability.
“Everyone should be doing this,” he said.
More to Read
Sign up for Essential California
The most important California stories and recommendations in your inbox every morning.
You may occasionally receive promotional content from the Los Angeles Times.









