A coronavirus immunity test is essential for the U.S. But will it work?

- Share via
It’s a potential saving grace salvaged from a pandemic: As people recover from the coronavirus, they may develop immunity that could allow them to return to school or work, helping place the U.S. on the road to recovery.
But tests to determine whether a person is immune are just being developed in the U.S., and concern is growing that the process could be marred by the same kind of widely criticized delays and confusion that plagued testing aimed at detecting COVID-19, the disease caused by the novel coronavirus.
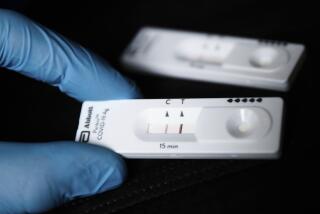
The serology tests use blood samples, rather than secretions taken from throats and noses, to check whether a person has developed antibodies against the coronavirus. Germany may begin widespread serology testing this month, and the U.K. has ordered millions of at-home antibody tests.
The U.S., by comparison, hasn’t come up with a cohesive plan for large-scale antibody testing, which health experts say could dash chances for a return to public life and leave health officials with few options for managing the pandemic other than severe social restrictions until a vaccine or drug therapy is available.
“If I were emperor and had unlimited resources, I’d do serological testing on every person in this country as fast as I could,” said Dr. David Merin, an emergency room physician with the Emergency Medicine Specialists of Orange County, who said he sees dozens of suspected COVID-19 patients each day and struggles to understand the delay in serological testing.
As the United States debates how long social distancing restrictions should stay in place, serology testing, if coupled with other forms of mitigation such as contact tracing, could provide relief by allowing those who have some immunity to return to work or school. Germany may issue “immunity certificates” allowing release from quarantine for those who show they have already been exposed and fought off the coronavirus.
If enough people in an area had immunity, whole neighborhoods or cities might see restrictions loosened. Or, in the case of healthcare workers and others on the front lines who lack protective gear, it could identify who has a lower risk if exposed.
Widespread serology testing would also give a more accurate picture of where the virus has been — and where it’s likely to go next.
Dr. Farzad Mostashari, a former federal health official who ran a door-to-door serological testing program for West Nile virus in the 1990s, said it’s possible that the pathogen has spread farther than realized in the United States and that many people have developed immunity. The opposite is also possible, that the novel coronavirus is just beginning its assault on the U.S.
“We don’t know right now which of those two scenarios are operating,” Mostashari said. Serological testing could illuminate “which of those two universes we are living in.”
The next testing challenge in the coronavirus pandemic is to diagnose those who were infected and then recovered. They are the ones with COVID-19 immunity.
The two federal agencies that would probably play roles in large-scale serology testing have been harshly criticized for botching the COVID-19 testing process.
The Centers for Disease Control and Prevention, which was slow to develop a coronavirus diagnostic test and later distributed a flawed one, is still in the initial stages of developing a serology test. Last month, CDC Director Dr. Robert Redfield told Congress the agency was developing two such tests, but neither has been released.
The Food and Drug Administration, which is responsible for vetting tests developed by others, was widely criticized for its unwillingness to speed up the approval process for diagnostic tests. But the agency has taken a nearly opposite approach to serological tests. It has waived many guidelines for bringing them to market and given permission for states to implement and manage their own testing without FDA input — as long as companies inform consumers they are not FDA approved. Under this lax scheme, it has allowed the sale of more than 40 serological tests, not requiring them to undergo a formal emergency-use approval process.
With regulatory hurdles removed, academic institutions, private labs and entrepreneurs are rushing to create their own blood tests or import them from abroad. But the accelerated process has created potential problems because there is no centralized collection of results, no uniformity of methods and slim evidence that the tests work with acceptable accuracy.
These tests run the gamut from consumer-oriented at-home models, similar to a pregnancy test and meant to help diagnose COVID-19, to lab-based tests designed to be performed by medical professionals with research in mind. The mix may confuse consumers who don’t understand the difference between having the infection and having immunity, and could give a false sense of security, experts said.
Even with antibodies present, no one knows how long immunity would last. It could range from a few months to years. The tests themselves are not all equal and do not check for the same markers, probably causing wide variation in what the results ultimately mean.
For public health officials, the myriad options may muddy the waters on the best way to move forward with broader testing if states or cities attempt their own serological surveys.
One of the most promising tests for public health was released last week by the Icahn School of Medicine at Mount Sinai, health experts said. Researchers there already posted the recipe for their antibody test to help other institutions jump-start their own efforts.
Near the other end of the spectrum, in the consumer market, Biomerica, an Irvine company, said it has shipped samples of its 10-minute disposable serology tests to the Middle East and Europe. The company plans to begin filling bulk orders overseas within weeks — but said it was awaiting “possible clearance and eventual use” domestically.
And Tuesday, a serology test aimed at an upscale market became available to anyone with $250 through NextHealth, a luxury wellness lounge where “health hackers” offer intravenous vitamin therapy and other elite services in West Hollywood.
Scientists racing to stave off a tidal wave of coronavirus infections show renewed interest in a little-known medicine: convalescent plasma.
Even as tests begin to hit the market, the U.S. government has only hinted that it’s planning the kind of wide serology study that could relieve social restrictions. No formal plans have been announced. But that comprehensive effort is necessary to gather the kind of data that will make serology results useful for resuming more normal life.
“You have to have a national database,” said Dr. Peter Beilenson, Sacramento County’s health officer. “It’s protecting the population. Whether it’s against war or viruses, that’s their role.”
Dr. Mark McClellan, who directs the Duke-Margolis Center for Health Policy and was the FDA commissioner from 2002 to 2004, said he believes the federal government is “making progress” on a surveillance program, but “we’re not there yet.”
“This is not over by a long shot,” he said. “The sooner we can take these additional steps on surveillance and serology … the sooner we can get back to the new normal.”