How does Britain’s Oxford-AstraZeneca vaccine compare?
- Share via
The U.K. approved a second COVID-19 vaccine for use in that country, and it’s a homegrown one this time. The government has ordered 100 million doses of the shot developed by AstraZeneca and the University of Oxford, more than any other candidate.
When will the vaccinations start?
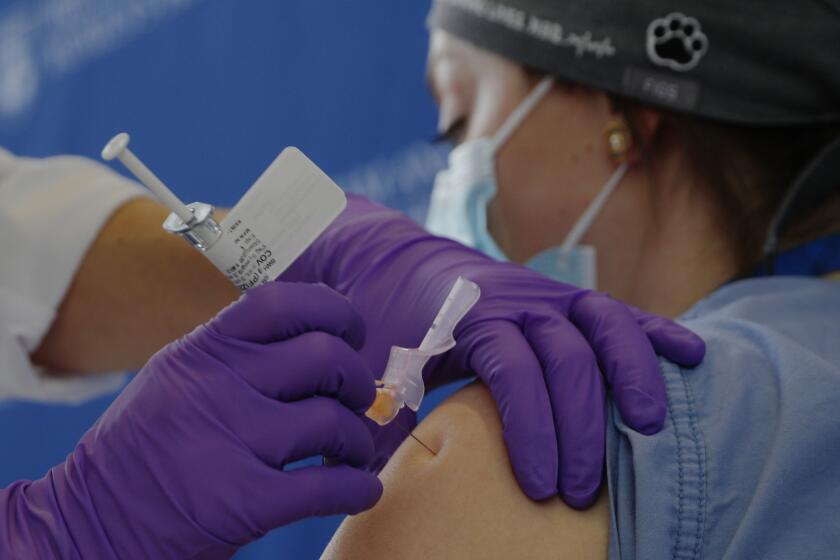
The first doses are being released in the U.K. Wednesday, and the vaccination campaign will start Monday. AstraZeneca says it aims to supply millions of doses in the first quarter.
For the record:
1:53 p.m. Dec. 30, 2020An earlier version of this story said the vaccine made by Pfizer and BioNTech turned cells into vaccine-making factories. The vaccine prompts cells to create a piece of the coronavirus to stimulate an immune response.
The government says the priority should be to give as many people in at-risk groups their first dose, rather than to provide the required two doses in as short a time as possible. But people should get the second shot four to 12 weeks after the first.
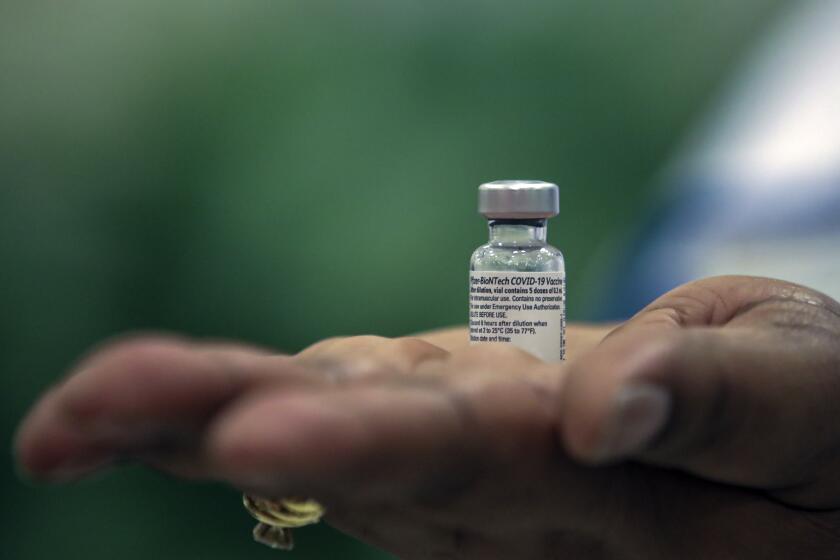
The first vaccine the U.K. approved, made by Pfizer Inc. and BioNTech, requires two injections three weeks apart.
Health experts hope to nudge people of color to the front of the COVID-19 vaccine line without explicitly saying race, ethnicity influence priority.
How does the vaccine differ from others?
The product uses a harmless chimpanzee virus to transport genetic material that triggers an immune response to the coronavirus. That’s different from the messenger-RNA approach used by Pfizer and BioNTech and by another vaccine made by Moderna that has been authorized for use in the United States. Those vaccines prompt the body to make a protein on the surface of the coronavirus, which in turn stimulates the immune system to make antibodies.
, which transforms the body’s own cells into vaccine-making factories.
When patients in a clinical trial were given two full doses of the Astra-Oxford vaccine, it was 62% effective — less than the Pfizer-BioNTech and Moderna vaccines.
If the COVID-19 vaccine from Pfizer and BioNTech was good enough to get a nod from the FDA, the vaccine from Moderna and the NIH almost certainly is as well.
A small group that mistakenly received a half-dose with the first shot showed better protection, with efficacy reaching 90%. But participants were 55 and younger, and because older people who are most at risk of severe cases of COVID-19 often show a more sluggish immune response, the results leave some doubt as to whether the higher efficacy will stand up to further testing.
Which dosage will be used?
The U.K. will administer the vaccine in two full doses rather than the half-dose, full-dose regimen. AstraZeneca has said it plans more clinical research to find out whether the results of the half-dose group hold up.
Why did the U.K. order more doses of this vaccine?
The Astra-Oxford shot is easier to transport and store: It can last six months at refrigerator temperatures, whereas the Pfizer-BioNTech vaccine requires deep freezing. The majority of doses will also be produced in the U.K., which should help avoid any supply delays.
Americans waiting their turn for a COVID-19 vaccine may not be pleased to see who’s in line ahead of them. Does a prisoner or smoker deserve it more?
Will the U.K. allow people to get both vaccines?
A regulatory panel advised against mixing doses of different vaccines because that combination hadn’t been tested. But
it’s possible the shots could be pooled in the future if studies show that can produce an enhanced immune response.
The U.K.’s Vaccine Taskforce has outlined plans to test combinations of approved shots next year to see if a mix could boost immunity, the panel said this month. The first tests will combine the Pfizer and Astra-Oxford vaccines.
A combination should work for vaccines that target the spike protein of SARS-CoV-2, according to Andrew Pollard, who led the University of Oxford’s vaccine trial with AstraZeneca.