Pfizer asks FDA to approve emergency use of its COVID-19 vaccine

- Share via
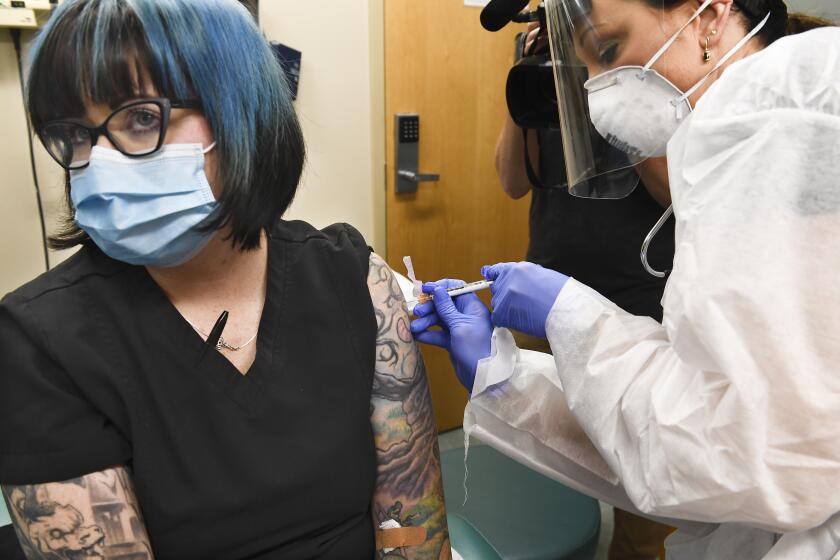
Pfizer formally asked U.S. regulators Friday to allow emergency use of its COVID-19 vaccine, starting the clock on a process that could bring limited first shots as early as next month and eventually an end to the pandemic — but not until after a long, hard winter.
The action comes days after Pfizer Inc. and its German partner BioNTech announced that its vaccine appears to be 95% effective at preventing mild to severe COVID-19 disease in a large, ongoing study.
The companies said that protection plus a good safety record means the vaccine should qualify for emergency use authorization, something the Food and Drug Administration can grant before the final testing is fully complete. In addition to the FDA submission, they have already started “rolling” applications in Europe and the U.K. and intend to submit similar information soon.
With the coronavirus surging around the U.S. and the world, the pressure is on for regulators to make a speedy decision.
“Help is on the way,” Dr. Anthony Fauci, the top U.S. infectious disease expert, said on the eve of Pfizer’s announcement, while adding that it’s too early to abandon masks and other protective measures. “We need to actually double down on the public health measures as we’re waiting for that help to come.”
Friday’s filing sets off a chain of events as the FDA and its independent advisors debate whether the vaccine is ready. If so, still another government group will have to decide how the initial limited supplies are rationed out to anxiously waiting Americans.
How much vaccine will be available and when is a moving target, but initial supplies will be scarce and rationed. Globally, Pfizer has estimated it could have 50 million doses available by year’s end.
About 25 million may become available for U.S. use in December, 30 million in January and 35 million more in February and March, according to information presented to the National Academy of Medicine this week. Recipients will need two doses, given three weeks apart. The U.S. government has a contract to buy millions of Pfizer-BioNTech doses, as well as other candidates that pan out, and has promised the shots will be free.
Not far behind is competitor Moderna Inc.’s vaccine. Early data suggest the shots are as effective as Pfizer’s, and the company expects to also seek emergency authorization within weeks.
There are still many unknowns when it comes to the pandemic, but the early signs of success for two experimental COVID-19 vaccines make a few things clear.
Here’s what happens next:
Making the data public
The public will get its first chance to see how strong the evidence of the vaccine’s effectiveness is in early December at a public meeting of the FDA’s scientific advisors.
So far, what’s known is based only on statements from Pfizer and BioNTech. Of 170 infections detected among trial subjects to date, only eight were among people who had received the vaccine; the rest had received a dummy shot. On the safety side, the companies cite results from 38,000 study participants who have been tracked for two months after their second dose. That’s a milestone set by the FDA because, historically, vaccine side effects don’t crop up later than that.
Coronavirus vaccines developed by Moderna and Pfizer show highly promising preliminary results in large-scale clinical testing. But key questions remain.
“We’ll drill down on these data,” said FDA advisor Dr. Paul Offit of the Children’s Hospital of Philadelphia.
A few days before meeting, the FDA will release its own internal analysis of the vaccine. That sets the stage for the advisors’ daylong debate about any signs of safety concerns and how the new vaccine technology works before they render a verdict.
They will recommend not just whether the FDA should allow broader use of the vaccine but also, if so, for whom. For example, is there enough proof the vaccine works as well for older, sicker adults as for younger, healthier people?
There’s still no guarantee. “We don’t know what that vote’s going to be,” said former FDA vaccine chief Norman Baylor.
Emergency use isn’t the same as full approval
If there’s an emergency green light, “that vaccine is still deemed investigational. It’s not approved yet,” Dr. Marion Gruber, chief of the FDA’s vaccine office, told the National Academy of Medicine this week.
That means that anyone offered an emergency vaccination must first receive a “fact sheet” describing potential benefits and risks before going through with the shot, she said.
The federal government has poured billions into COVID-19 vaccine research, but will Americans reap the profits?
There will be a lot of unknowns. For example, the 95% protection rate is based on people who developed symptoms and then were tested for the virus. Can a vaccinated person get infected but have no symptoms and then spread the coronavirus? How long does protection last?
That’s why the 44,000-person study needs to keep running.
At least for now, pregnant women won’t qualify because they weren’t studied. Pfizer only recently began testing the vaccine in children as young as 12.
A decision on Pfizer’s vaccine won’t affect other COVID-19 vaccine candidates in the pipeline, which will be judged separately.
Manufacturing
Brewing vaccine is more complex than typical drug manufacturing, yet the millionth dose to roll out of Pfizer’s Kalamazoo, Mich., factory must be of the same purity and potency as every dose before and after.
That means the FDA decision isn’t based just on study data but on its determination that the vaccine is being made correctly.
The Pfizer-BioNTech vaccine — and Moderna’s candidate — are made with brand-new technology. They don’t contain the actual coronavirus. Instead, they’re made with a piece of genetic code for the “spike” protein that studs the virus.
That messenger RNA, or mRNA, instructs the body to make some harmless spike protein, which then trains immune cells to recognize the real coronavirus if it comes along.
Getting doses out to people
Another government group, advisors to the Centers for Disease Control and Prevention, decides who is first in line for scarce doses. Health and Human Services Secretary Alex Azar said he hopes that decision can be made at the same time as the FDA’s.
California and other states are racing to finalize plans for who will get the first doses of COVID-19 vaccines and how they will be delivered.
The Trump administration’s Operation Warp Speed has worked with states to line up how many doses they would need to cover the populations offered the vaccine first.
Pfizer will ship those supplies as ordered by the states only after the FDA gives the OK.
Company projections of how much will be shipped each month are just predictions, Baylor warned.
“It’s not like a pizza,” he said. Manufacturing is so complex that “you don’t necessarily end up with what you thought.”
More to Read
Sign up for Essential California
The most important California stories and recommendations in your inbox every morning.
You may occasionally receive promotional content from the Los Angeles Times.