Beware of buying young people’s blood to prevent aging, FDA says

- Share via
Taking a young person’s blood plasma and infusing it into an older person to ward off aging — a therapy that’s fascinated some of the biggest names in Silicon Valley — has no proven clinical benefit, the Food and Drug Administration said.
The agency issued a safety alert Tuesday about the infusion of plasma from young donors for the prevention of conditions such as aging or memory loss, or for the treatment of such conditions as dementia, Parkinson’s disease, multiple sclerosis, Alzheimer’s disease, heart disease or post‐traumatic stress disorder.
“There is no proven clinical benefit of infusion of plasma from young donors to cure, mitigate, treat or prevent these conditions, and there are risks associated with the use of any plasma product,” the FDA said in a statement from Commissioner Scott Gottlieb and Peter Marks, head of the agency’s biologics center.
The idea of infusing young blood to fight aging has attracted technology entrepreneurs such as billionaire Peter Thiel and was lampooned in a 2017 episode of the HBO show “Silicon Valley.”
Thiel’s reported interest was sparked by a company called Ambrosia, whose website said Tuesday morning that the company had locations in five U.S. states — including a Los Angeles office — and was selling 1 liter of blood plasma, from donors ages 16 to 25, for $8,000. By Tuesday afternoon, Ambrosia’s website said that because of the FDA announcement the company had “ceased patient treatments.”
Gottlieb and Marks said none of the plasma treatments had gone through the rigorous testing required by the agency. Ambrosia says “experiments in mice called parabiosis provided the inspiration to deliver treatments with young plasma.” The FDA typically requires human trials before companies can make a specific health claim about a product.
“The reported uses of these products should not be assumed to be safe or effective,” Gottlieb and Marks said. “We strongly discourage consumers from pursing this therapy outside of clinical trials under appropriate institutional review board and regulatory oversight.”
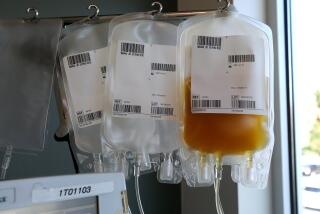
Plasma, the liquid portion of blood, contains proteins to help the blood clot. Plasma infusion is an approved use by the FDA in trauma settings or in patients whose blood doesn’t coagulate. But, the FDA says, there are risks, including allergic reactions, circulatory overload, lung injury and transmission of infectious diseases.
More to Read
Inside the business of entertainment
The Wide Shot brings you news, analysis and insights on everything from streaming wars to production — and what it all means for the future.
You may occasionally receive promotional content from the Los Angeles Times.