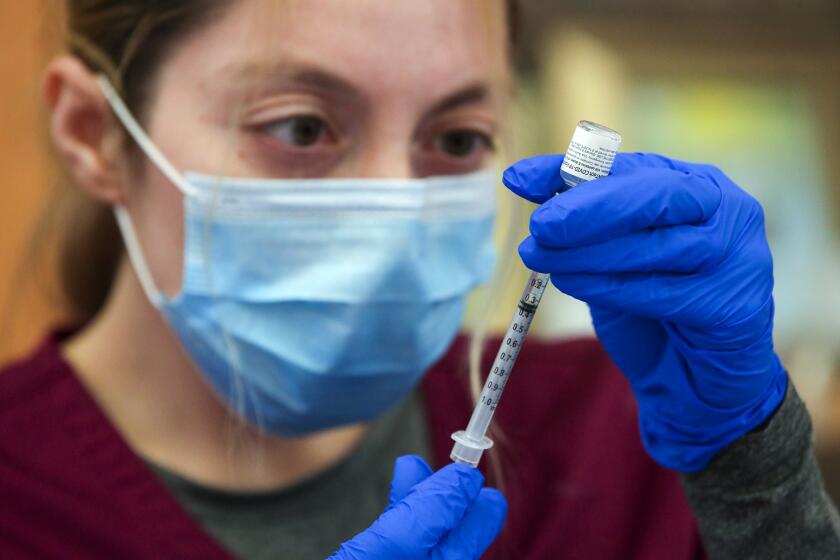
Immunocompromised people shouldn’t have to wait for COVID-19 booster shots
- Share via
Last week, Pfizer announced that it plans to request an emergency use authorization for a booster vaccine shot for COVID-19. Within hours, the Food and Drug Administration and the Centers for Disease Control and Prevention issued a joint statement asserting, “Americans who have been fully vaccinated do not need a booster shot at this time.” The Department of Health and Human Services reiterated this after it and senior U.S. scientists met with top Pfizer representatives this week. Immunologists, health policy experts, the director of the World Health Organization and pundits have criticized Pfizer for planning to request this approval. Some suggest that it’s a cash grab for the vaccine maker and others assert simply that more data are needed.
What these responses ignore is growing evidence that a third shot may provide lifesaving protection to immunocompromised people (including organ transplant patients), who represent approximately 3% to 4% of our adult population.
The evidence is clear: Two doses of Pfizer’s COVID-19 vaccine is highly protective for most people. But there are subpopulations for whom these powerful vaccines are demonstrably not yet working. It is likely that hundreds of thousands of fully vaccinated individuals in the U.S., if not more, remain at just as much risk of contracting COVID-19 as they were before they were vaccinated. Many of them would likely benefit greatly from a booster shot — but despite the surplus of vaccine in the United States at this point, they can’t currently get that booster because it’s not yet authorized.
We know, because one of us fits squarely in that group.
Last December, one of us, Jennifer, donated a kidney to the other, her father, Robert, who had end-stage kidney disease. Surgery at UCLA removed Jennifer’s left kidney, which was then flown on a red-eye to Boston and transplanted into Robert at Massachusetts General Hospital the next morning.
The good news is that the surgery was successful. The bad news is that the two doses of Pfizer vaccine that he got a few months later seem to have failed to work.
More than a month after his second jab, Robert’s blood was tested for the presence of the antibodies the vaccine generates in most people. His antibody level came back at zero — completely undetectable. While it’s not completely certain that a failure to detect antibodies means zero protection against COVID-19, Robert’s doctors suggest that’s likely.
There’s a sensible medical explanation for this. As a transplant recipient, Robert takes powerful immunosuppressant drugs. Those drugs make him more susceptible to infection of all kinds, including COVID-19. They also — by design — make it harder for his body to generate any immune response. That’s a huge plus for his body’s response to Jennifer’s “foreign” kidney, but it’s likely the reason why his two vaccine doses failed to spark the immune response that generates protection.
Robert’s situation is not unusual. A Johns Hopkins study of transplant recipients and other immunocompromised individuals showed that nearly half had no immune response to two doses of a COVID mRNA vaccine. This indicates that a substantial portion of immunocompromised Americans currently lack all protection even if vaccinated. This is a group at heightened risk for serious COVID-19 given the underlying health conditions that make them immunocompromised.
In a promising recent development a French study showed that nearly half of test subjects in parallel situations responded well to a booster shot. Among the 59 patients who lacked antibody response after two shots, 44% showed positive results after a third shot, with no serious adverse events. An observational study from Johns Hopkins that looked at a small set of American transplant patients who finagled a third shot even though it’s not yet authorized also showed significant promise. France and Israel have already approved boosters for immunocompromised people and the United Kingdom has announced that it plans to give boosters to immunocompromised people and older people starting this fall.
But Robert’s doctors have told him that they do not have the authority to recommend a third shot right now.
This is because the emergency use status for an approved two-dose vaccine protocol provides no leeway for doctors to recommend a booster. The CDC’s lawyers advised a Massachusetts research hospital that administering a third dose would be a violation of the CDC “Provider Agreement,” because the emergency authorization did not explicitly provide for it. Worse still, we are told by Robert’s doctors that there are substantial regulatory barriers preventing American medical researchers from pursuing formal studies to test giving boosters to immunocompromised patients.
Meanwhile, the highly contagious Delta variant poses additional risk for this group, especially given our collective move away from masking and social distancing. If mild breakthrough cases are more frequent for this new variant, vaccinated-but-immunocompromised people cannot protect themselves by limiting their interactions to those who are vaccinated.
At a bare minimum, the CDC should encourage clinical trials of boosters for transplant patients and immunocompromised people and remove the existing barriers to such studies. We would go even further and encourage the CDC to follow the French and Israeli examples and permit a booster now for this population.
Two other broader solutions would also open the door for immunocompromised people to access booster shots. If the FDA grants full approval to the vaccines, doctors will have more flexibility to use their clinical judgment about the wisdom of an “off label” use of a booster. And if Pfizer receives emergency use authorization for a booster shot, that would also give immunocompromised people access.
As the Delta variant threatens to produce another wave of COVID-19 cases, we should not allow immunocompromised Americans to go unprotected when there is an easy solution: Allow a third shot now for those who need it.
A small study offers a hint that an extra dose of COVID-19 vaccine might give organ transplant recipients a boost in protection.
Jennifer Mnookin is dean of the UCLA School of Law. Robert Mnookin, her father, is a professor of law at Harvard.
More to Read
A cure for the common opinion
Get thought-provoking perspectives with our weekly newsletter.
You may occasionally receive promotional content from the Los Angeles Times.