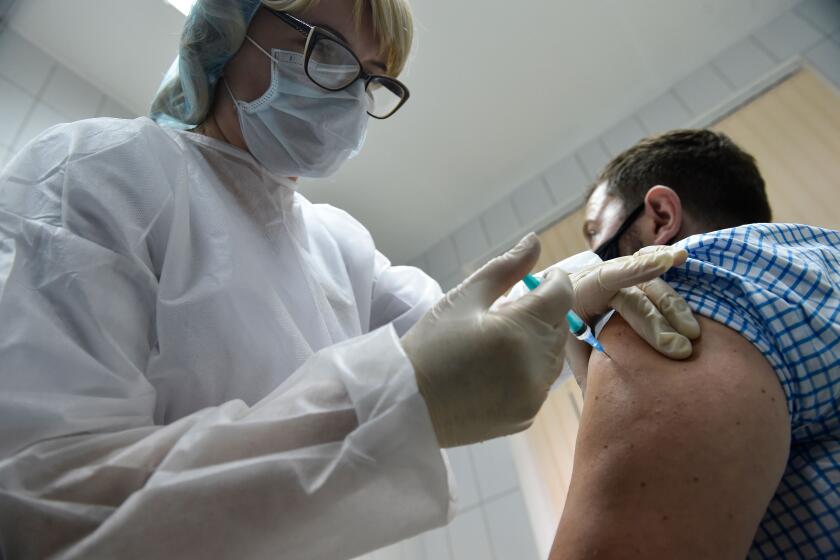
Putin orders ‘large-scale’ COVID-19 vaccination in Russia with locally made vaccine

- Share via
MOSCOW — Russian President Vladimir Putin has ordered a “large-scale” COVID-19 immunization campaign to start by late next week, with doctors and teachers set to be first in line to get a Russian-designed vaccine that has yet to complete the advanced studies needed to ensure its effectiveness and safety in line with established scientific protocols.
Putin’s action Wednesday came hours after Britain became the first country in the West to authorize the use of a COVID-19 vaccine, one developed by Pfizer and Germany’s BioNTech.
Russia touted its domestically developed vaccine, Sputnik V, as the world’s “first registered COVID-19 vaccine” after the government gave it regulatory approval in early August. However, giving the shots the go-ahead drew considerable criticism from experts, because at the time the Sputnik V had been tested on only several dozen people.
The Russian leader said at the time that the early vaccine recipients included one of his daughters.
Putin said Wednesday that more than 2 million doses of the Sputnik V shot have “been produced or will have been produced in the next few days.”
“This gives us the opportunity to start ... large-scale vaccination, and of course, as we agreed, first of all of the two risk groups — doctors and teachers,” Putin told government officials. He tasked Deputy Prime Minister Tatyana Golikova to “organize the work in such a way so that large-scale vaccination starts by the end of next week.”
Britain becomes the first country to approve a fully tested COVID-19 vaccine, but its show of national pride raises EU hackles.
Golikova said that the shots would be voluntary and free of charge, and that the government was looking to include other demographic groups in the vaccination campaign.
The two-shot Sputnik V vaccine was developed by the Moscow-based Gamaleya Institute. An advanced study among 40,000 volunteers was announced two weeks after the vaccine received government approval. The trial is still ongoing.
But the vaccine is already being offered to people in high-risk groups such as medical workers, despite expert warnings against its wider use before all the necessary testing is done. Several high-profile officials have said they received shots.
Health Minister Mikhail Murashko said Wednesday that more than 100,000 people in Russia have been given the shots.
Touting a Russian vaccine is ‘huge gamble’ for Putin, but could carry rewards as well
Kirill Dmitriyev, head of the Russian Direct Investment Fund that bankrolled Sputnik V’s development, added that about 25,000 people have been vaccinated with both doses of the vaccine “as part of a clinical trial now.” Dmitriyev said that outside of the trial, the vaccine was offered to “volunteers” among medical workers “because we wanted to protect our front-line personnel first.”
Last month, developers of the vaccine said interim analysis of trial data showed it was 91.4% effective.
Dmitriyev said Wednesday that developers of the vaccine have applied to the European Medicines Agency to get Sputnik V approved for use in Europe, and submitted applications in other countries.
“We understand that it may take some time, but we concurrently, you know, have applied for approval in 40 different countries and we are working very closely with regulators in all of those countries,” Dmitriyev said.
It could be a while before U.S. efforts to develop coronavirus vaccines benefit the developing world. China and Russia are trying to fill the void.
Dmitriyev said last month that more than 1 billion doses of the vaccine were expected to be produced next year outside Russia. Putin’s spokesman, Dmitry Peskov, however, stressed Wednesday that vaccinating Russians “will be the absolute priority.
Russia’s total of more than 2.3 million confirmed cases is currently the fourth-largest caseload in the world. On Wednesday, officials reported 25,345 new cases.
More to Read
Sign up for Essential California
The most important California stories and recommendations in your inbox every morning.
You may occasionally receive promotional content from the Los Angeles Times.