Nearly 160 coronavirus vaccines are in the works. Here’s a closer look at the science

- Share via
In the world of virology, the nomenclature of war comes easy. The human body is a citadel that relies on the immune system to defend it. A virus is an attacking army that does everything it can to overrun those defenses.
Vaccines help. Though they do not prevent the virus from causing an infection, they can control the infection before it leads to symptoms and disease. By staging a scrimmage against the immune system, vaccines teach the body to enlist and train a specialized force of white blood cells and antibodies that are called up in the event of a life-threatening attack.
With more than 7.5 million confirmed cases worldwide, the new coronavirus has killed more than 420,000. Creating vaccines to protect humans against this threat is a top priority.
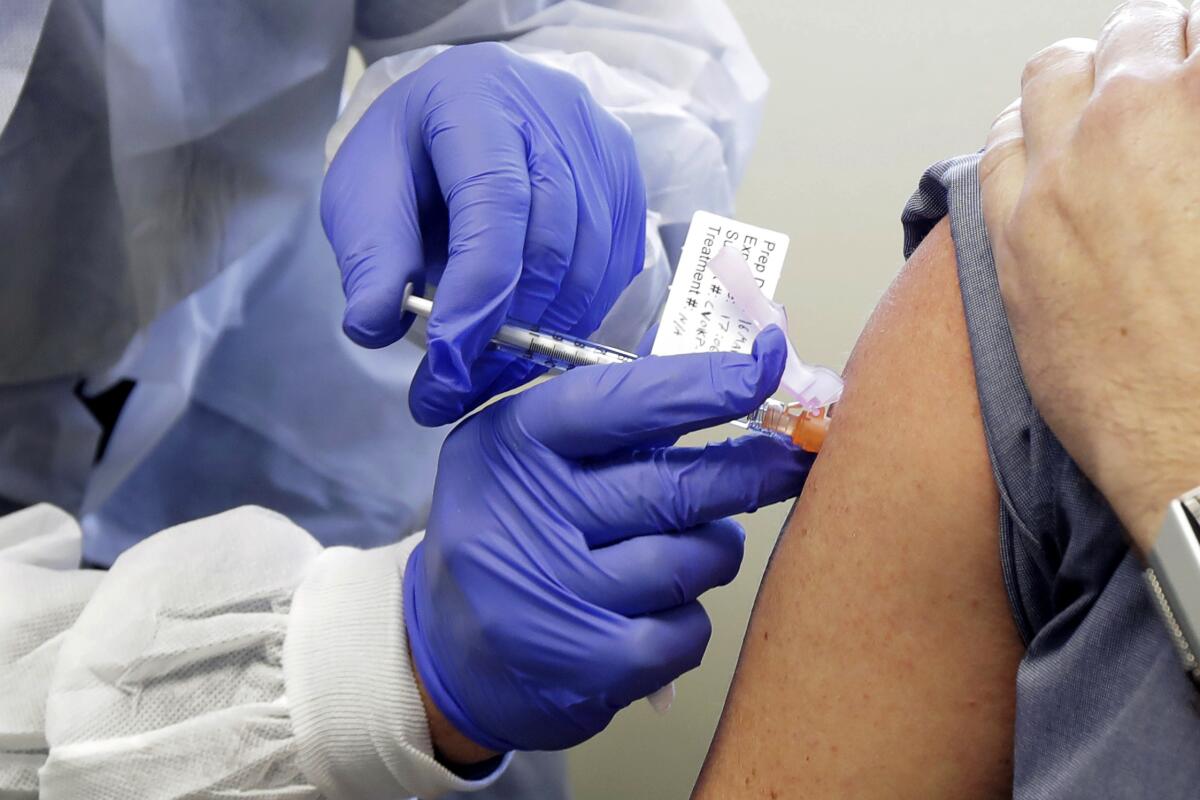
Nearly 160 COVID-19 vaccine candidates are in development in laboratories around the world. The U.S. Department of Health and Human Services has singled out five contenders: Moderna; a consortium of Oxford University and AstraZeneca; Johnson & Johnson; Merck; and Pfizer.
Although all approaches are distinct, they are based on a few simple strategies. Some have been around for years; others are being tested for the first time.
Here’s a closer look at how they work and which laboratories are farthest along in their development.
Traditional approaches
Some researchers are attempting to use the entire SARS-CoV-2 coronavirus to create vaccines that will induce a broad immune response. They are borrowing techniques developed over centuries by physicians and healers who knew that minor viral infections can inoculate patients against more serious disease.
An advantage of these methods is their proven efficacy. They have helped defeat polio, hepatitis A, rabies and other diseases. At least 12 laboratories are working to add COVID-19 to this list.
But these vaccines are challenging to create, and production is time-consuming. They are often grown in chicken eggs, so millions of hens could be needed to mass-produce a vaccine for worldwide distribution, and working with a live virus is risky.

Live attenuated virus vaccine
Developed in 1937 by virologist Max Theiler to create a yellow fever vaccine, this protocol takes a live virus and introduces it to mice, chicken embryos or other non-human species. As the virus becomes more successful at replicating in nonhuman cells, it loses its ability to replicate in human cells.
However, when introduced back into the human body, attenuated viruses still trigger an attack by the immune system, which is then able to stave off disease in most cases.
Vaccines developed from attenuated viruses provide protection against measles, mumps, rubella, chicken pox and one type of rotavirus.
This protocol was used by Albert Sabin to create a polio vaccine that could be administered orally in a sugar cube. Because it doesn’t need to be injected, it is a less expensive way to inoculate people around the world.
No laboratories are currently in clinical trials with a live attenuated virus vaccine.
Inactivated virus vaccine
This protocol, pioneered by Jonas Salk in the early 1950s, takes live viruses and kills them so they can’t replicate. The inactivated, or dead, virus is injected into the body, which prompts the creation of antibodies.
Because the entire virus is used, the body mounts a broad immune response that is not as robust as with vaccines made from live attenuated viruses. Inactivated virus vaccines often require periodic booster shots to re-stimulate the immune system.
This protocol was used to create Salk’s injectable polio vaccine, as well as the hepatitis A and rabies vaccines.
Four teams have advanced inactivated virus vaccines to clinical trials: the Wuhan Institute of Biological Products and Sinopharm; the Beijing Institute of Biological Products and Sinopharm; the Institute of Medical Biology, Chinese Academy of Medical Sciences; and Sinovac.Newer approaches
Rather than use the entire coronavirus to induce an immune response, many researchers are hoping that a single protein on its surface can do the job.
The so-called spike protein, or S protein, plays an essential role in allowing the virus to bind and infiltrate a host cell and appropriate the genetic material it needs to replicate. Disable the S protein, the theory goes, and the virus can’t access the host cell.
More than 100 laboratories are focused on stopping the S protein. While technically challenging, this method for developing a vaccine might be easier and less expensive to produce than using traditional approaches.
But some researchers have expressed concerns that if the S protein mutates, a vaccine that targets it will be ineffective.
Replicating and non-replicating viral vector vaccine
Researchers begin by splicing genetic instructions for making the S protein into a harmless virus, which is then injected into the body. The virus uses human cells to mass-produce the protein, and the immune system attacks both the virus and the protein with antibodies.
Sometimes the harmless virus has been modified so that it cannot replicate inside the body. Non-replicating viruses might require higher doses or booster shots. Replicating viruses make more of the S protein as they infect more cells.
Researchers used this approach to develop an Ebola vaccine.
Two research groups have advanced non-replicating viral vector vaccine to clinical trials: Oxford University and AstraZeneca, and CanSino Biological and the Beijing Institute of Biotechnology.
Two others are said to be close to starting their clinical trials: the Johnson & Johnson subsidiary Janssen, and NantKwest and ImmunityBio. (Dr. Patrick Soon-Shiong, owner of The Times, is chairman and CEO of NantKwest and ImmunityBio.)
The pharmaceutical company Merck, working with the nonprofit research organization IAVI, is said to be close to starting a clinical trial with a replicating viral vaccine.
Virus-like particle vaccine
Rather than teach the body to make antibodies against the S protein, researchers are trying to develop a “particle” that resembles the coronavirus on the outside but without the deadly genetic machinery on the inside. Once introduced into the body, this harmless particle should appear threatening and foreign enough for the immune system to initiate an assault.
The human papillomavirus vaccine takes this approach. It is believed to be effective because it is based not just on a single protein but a three-dimensional representation of the entire virus.
No laboratories are currently in clinical trials with a virus-like particle vaccine.
Protein-subunit vaccine
Researchers hope that by injecting synthetically produced S protein — or parts of the S protein — into the body with no genetic material attached, they can induce an immune response that would prime the body to attack the coronavirus. This type of vaccine does not require a virus to deliver the protein.
If this defense isn’t entirely successful, it could still prevent an infection from developing serious complications like pneumonia. Some influenza vaccines work this way.
Of all the methods being tracked, both new and old, protein-subunit vaccines are the most common and are currently being developed in 51 laboratories. Only one laboratory has advanced this method to a clinical trial: Novavax.
RNA- and DNA-based vaccines
Researchers have sequenced the SARS-CoV-2 genome and are using it to synthesize the virus’ RNA. Like many viruses, coronaviruses have RNA instead of DNA, and in some laboratories, scientists use the RNA to create the corresponding DNA.
Once the genetic material is injected into the body — without a virus to carry it — it is taken up by cells that begin producing the S protein, triggering an immune response.
RNA- and DNA-based vaccines have not yet been tested on humans, although a gene therapy treatment for infants with spinal muscular atrophy uses a similar approach.
There is some concern that the vaccine’s DNA could be incorporated into the body’s cells and subvert the cell’s normal function. But DNA has the advantage of being more stable than RNA, which readily degrades.
Thirty-three laboratories are taking this approach. One has advanced a DNA-based vaccine to a clinical trial: Inovio Pharmaceuticals. Two other laboratories have reached the clinical trial stage with an RNA-based vaccine: Moderna; and BioNTech, Fosun Pharma and Pfizer.
Sources: Paul Offit, Children’s Hospital of Philadelphia, University of Pennsylvania; Brenda Huneycutt, FasterCures, Milken Institute; Helen Keipp Talbot, National Foundation for Infectious Diseases, Vanderbilt University
More to Read
Sign up for Essential California
The most important California stories and recommendations in your inbox every morning.
You may occasionally receive promotional content from the Los Angeles Times.