California remains optimistic on COVID-19 recovery despite pausing J&J vaccine use
- Share via
California officials said Tuesday they were optimistic they could still hit vaccination and reopening targets even as they moved to temporarily stop administering the Johnson & Johnson COVID-19 vaccine.
The move came on the recommendation of federal health officials following reports of six serious blood clots nationwide. Officials characterized the stoppage as a cautionary move and emphasized that the reactions that prompted it are exceedingly rare.
The pause in J&J vaccines will not change California’s plan to expand vaccine eligibility to all residents 16 and older starting Thursday, or to fully reopen the economy two months from now, officials said.
“It will not materially impact our ability to fulfill our expectations and commitment to ... fully vaccinate all those that seek to get vaccinated so that we can begin to more fully open our economy by June 15,” Gov. Gavin Newsom said Tuesday during a news conference in Butte County, where he signed a bill to dedicate $536 million to wildfire prevention.
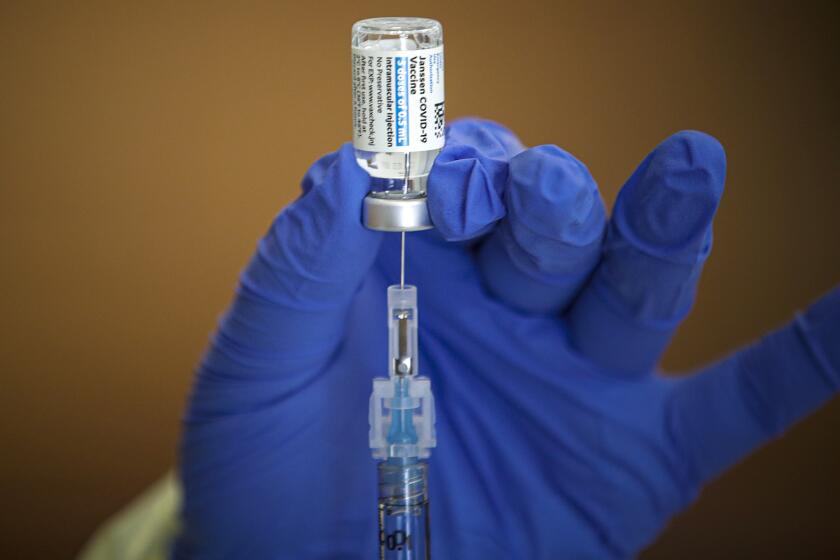
U.S. officials called for a pause in the use of Johnson & Johnson’s COVID-19 vaccine after serious blood clots were reported in six recipients.
Almost 875,000 Johnson & Johnson doses have been administered in California, according to data from the Centers for Disease Control and Prevention. Nationwide, the number is in excess of 7.2 million.
“The J&J vaccine has been extraordinarily safe, save for six specific reported instances that the CDC and the FDA have highlighted today,” Newsom said. “That’s less than one in a million incidences that were deemed severe.”
Officials also expressed confidence that the pause won’t throw a significant wrench in the state’s race to widely vaccinate as many people as possible. Less than 4% of California’s allocation from the federal government this week — 67,600 out of roughly 2 million doses — is from Johnson & Johnson.
The remainder is Pfizer-BioNTech and Moderna vaccines, both of which are being distributed and administered as normal.
“As the federal government has said, we do not expect a significant impact to our vaccination allocations,” state epidemiologist Dr. Erica Pan said in a statement.
Distribution of the Johnson & Johnson COVID-19 vaccine had been paused in California as more residents become eligible.
Newsom, who himself received the Johnson & Johnson vaccine April 1, said roughly 8,800 appointments scheduled for that vaccine through the state’s My Turn platform will need to be converted to Moderna and Pfizer alternatives.
“We’re mindful that, with the J&J, our ability to do as much as we anticipated this week, and over the next few weeks, is impacted,” he said. “But our medium- and long-term goals are not impacted because of the abundance of Moderna and Pfizer vaccines that we indeed anticipate not only this week, compared to next week, but also in the subsequent and upcoming weeks.”
In Los Angeles, the pause will not affect permanent vaccination sites, but will alter plans to provide about 3,000 scheduled doses through the city’s mobile MOVE clinics, officials said Tuesday.
“All appointments at our permanent sites will happen as planned, and the city’s MOVE clinics will honor the appointments for anyone scheduled to receive the Johnson & Johnson vaccine with a first dose of Pfizer as we await further information from health officials,” Mayor Eric Garcetti’s office said in a statement.
Before the state’s announcement, both the city and county of Los Angeles confirmed they, too, would halt use of the Johnson & Johnson shot.
County health officials said the pause would last until the Food and Drug Administration and CDC finished looking into the matter, a review that’s “expected to take several days.”
In the meantime, providers in the county “will contact patients about rescheduling or providing a new appointment for Pfizer or Moderna vaccine,” the Department of Public Health wrote in an email following an inquiry from The Times.
“People who received the vaccine in the last three weeks should look for any symptoms of these unusual clots, including severe headaches, abdominal or leg pain, and shortness of breath, and contact their medical provider if symptoms develop,” county officials wrote in their statement. “People who don’t have a medical provider can call 2-1-1 to connect with a healthcare provider.
“We are grateful to the researchers and scientists working to ensure that all medications or vaccines, including the COVID-19 vaccines, have the highest safety standards.”
We explain what vaccine passports are, how they work, where they’ve been implemented, and why some people object to them.
The CDC has scheduled an emergency meeting for Wednesday to discuss the Johnson & Johnson vaccine.
“Until that process is complete, we are recommending a pause in the use of this vaccine out of an abundance of caution,” said a joint statement from Dr. Anne Schuchat, principal deputy director of the CDC, and Dr. Peter Marks, director of the Food and Drug Administration’s Center for Biologics Evaluation and Research. “This is important, in part, to ensure that the healthcare provider community is aware of the potential for these adverse events and can plan for proper recognition and management due to the unique treatment required with this type of blood clot.”
Dr. Jeff Goad, professor and chair of the department of pharmacy practice at Chapman University, said the FDA panel would examine medical data to determine whether the small number of adverse reactions seem to be connected to the vaccine or are coincidental.
What people need to do, he said, “is wait and let the science guide and see what comes out of the discussions.”
“That’s why we have these safety systems in place, to very rapidly and very early on detect and protect people,” Goad said. “The systems are working.”
The Western States Scientific Safety Review Workgroup — which includes public health experts from California, Nevada, Oregon and Washington — planned to meet Tuesday to review the decision from the CDC and the FDA to pause the vaccine.
Are vaccines safe? When can I get a booster dose? Do I have to wear a mask? We’ve answered hundreds of newsletter readers’ questions. Here’s an archive.
While it’s unclear how long the Johnson & Johnson vaccine will remain on ice, any prolonged interruption threatens to become another speedbump in the road to ending the COVID-19 pandemic.
California officials had said that hitting the target date of June 15 for fully reopening the state economy would hinge heavily on vaccination progress.
To date, providers throughout California have administered 23 million COVID-19 vaccine doses, and 38.8% of residents have received at least one shot, according to figures from the CDC.
Even before the pause, California was running into supply issues with that vaccine.
Although California’s allocations of the Pfizer-BioNTech and Moderna vaccines are expected to remain relatively steady this week, the state — along with the rest of the nation — saw its supply of Johnson & Johnson slashed.
Last week, 574,900 Johnson & Johnson doses were allocated to the Golden State. This week, that number will plummet to 67,600, an 88% drop, CDC data show.
The allocation is expected to fall even further next week, to 22,400 doses, according to the California Department of Public Health.
The dropping number of available Johnson & Johnson doses stems from a production issue at a plant in Baltimore — where the drugmaker said a number of doses (news reports pegged the number at 15 million) failed quality standards and couldn’t be used.
Johnson & Johnson is now “working closely with the [Food and Drug Administration] to resolve any manufacturing issues,” as well as “installing a new senior leadership team to oversee all aspects of production and manufacturing at the facility,” according to Jeff Zients, coordinator of President Biden’s COVID-19 task force.
The dip will drive down the size of the state’s federal allocation from the 2.4 million doses received last week to 2 million this week and 1.9 million next week.
Chris Megerian contributed to this report.
More to Read
Sign up for Essential California
The most important California stories and recommendations in your inbox every morning.
You may occasionally receive promotional content from the Los Angeles Times.