Pfizer says further data show its COVID-19 vaccine is 95% effective, works in older people
- Share via
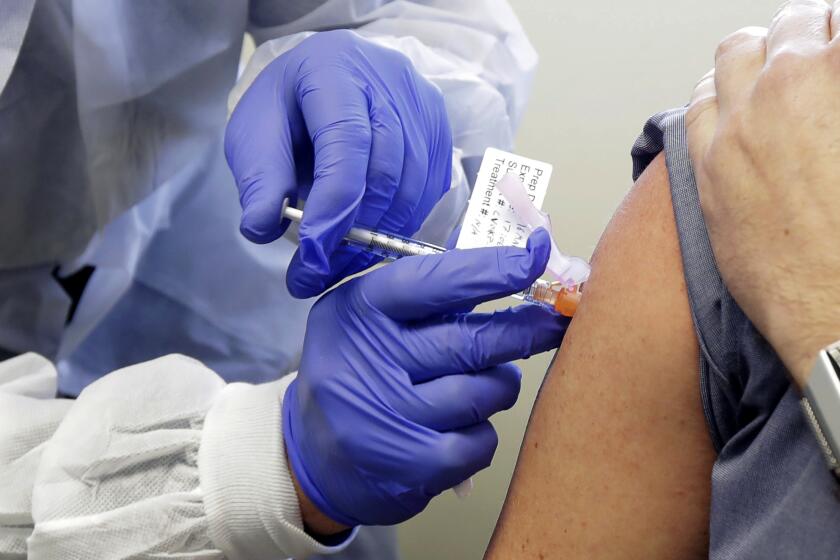
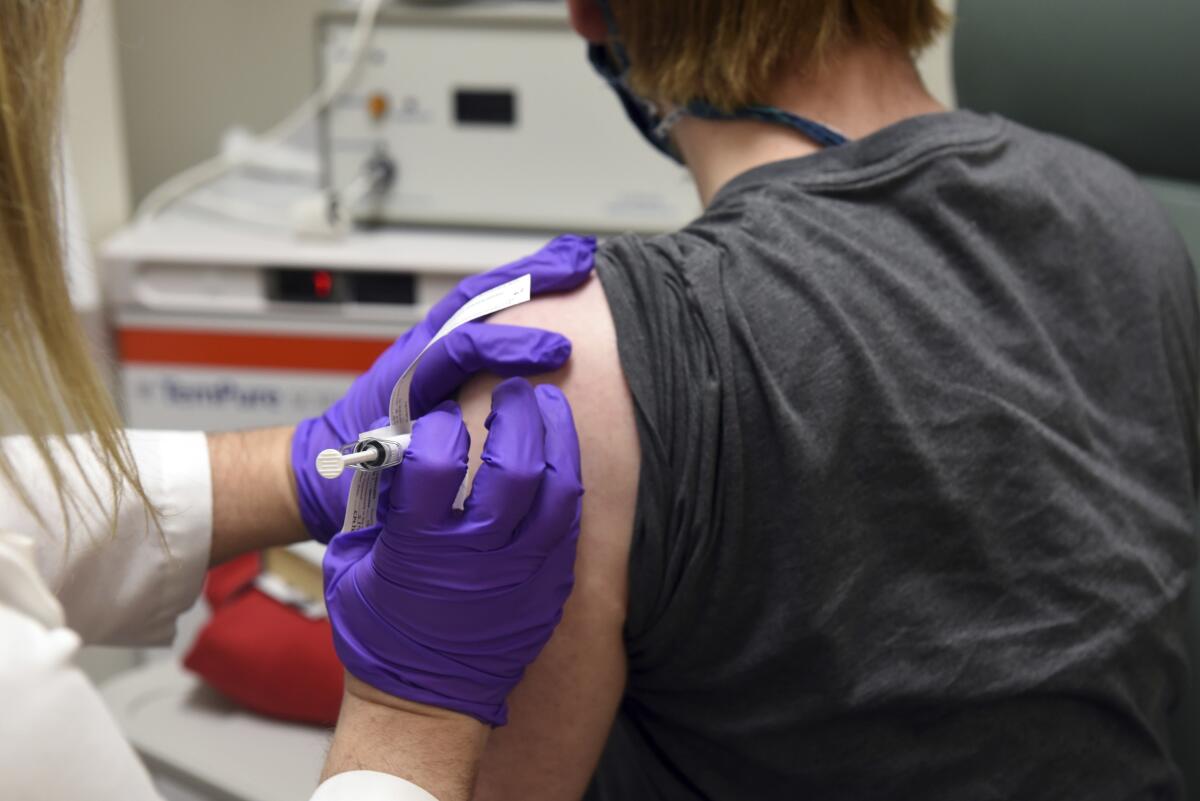
Pfizer said Wednesday that new test results show its COVID-19 vaccine is 95% effective, has no serious side effects and protects older people most at risk of dying of the disease — the last data needed for the company to seek approval for emergency use.
The announcement, just a week after Pfizer first revealed promising preliminary results, comes as the company is preparing within days to formally ask U.S. regulators to allow emergency use of the vaccine.
Pfizer and its German partner, BioNTech, also have begun “rolling submissions” for the vaccine with regulators in Europe, Britain and Canada, and soon will add this new data.
Pfizer and BioNTech initially had estimated that their vaccine was more than 90% effective after 94 infections had been counted. With Wednesday’s announcement, the companies now have accumulated 170 infections in the study — and said that only eight of them occurred in volunteers who got the actual vaccine rather than a dummy shot. One of those eight developed severe disease, the companies said.
“This is an extraordinarily strong protection,” Ugur Sahin, BioNTech’s CEO and co-founder, said.
The companies have not yet released detailed data on their study, and results have not been analyzed by independent experts. Also still to be determined are important questions such as how long protection lasts and whether people might need boosters.
Coronavirus vaccines developed by Moderna and Pfizer show highly promising preliminary results in large-scale clinical testing. But key questions remain.
The companies said their vaccine was more than 94% effective in adults over 65, although it is not clear how they determined effectiveness in older adults, with only eight infections to analyze in the vaccinated group and no breakdown provided of those people’s ages.
Sahin said there were enough older adults enrolled in the study and among the placebo recipients who became infected that he is confident “this vaccine appears to work in the higher-risk population.”
Earlier this week, Moderna announced that its experimental vaccine appears to be 94.5% effective after an interim analysis of its late-stage study.
Both vaccine candidates are made with a brand-new technology that uses a snippet of the coronavirus’ genetic code to train the body to recognize if the real virus comes along. The similar results of the two studies are likely to give further reassurance to experts about the novel approach.
While initial supplies will be scarce and rationed, Sahin said that, as supplies grow, the companies have a responsibility to help ensure access for lower-income countries as well.
Pfizer and BioNTech say they now have the data on the vaccine’s safety needed to seek emergency authorization from the Food and Drug Administration.
The companies didn’t disclose safety details but said no serious side effects have been reported, with the most common problem being fatigue after the second vaccine dose, affecting about 4% of participants.
The study has enrolled nearly 44,000 people in the U.S. and five other countries. The trial will continue to collect safety and efficacy data on volunteers for two more years.
Pfizer and BioNTech said they expect to produce up to 50 million vaccine doses globally in 2020 and up to 1.3 billion doses in 2021.
U.S. officials have said they hope to have about 20 million vaccine doses each from Moderna and Pfizer available for distribution in late December. The first shots will be offered to vulnerable groups such as medical and nursing-home workers and people with serious health conditions.
More to Read
Sign up for Essential California
The most important California stories and recommendations in your inbox every morning.
You may occasionally receive promotional content from the Los Angeles Times.