Full Coverage: Superbug outbreaks tied to faulty scopes
A number of U.S. hospitals have experienced bacterial outbreaks related to contaminated medical scopes, including at UCLA’s Ronald Reagan Medical Center and Cedars-Sinai hospital.
- 1
A U.S. lawmaker is renewing his push for Congress to toughen requirements on medical-device warnings, calling Olympus Corp.’s 2013 decision against issuing a broad alert to U.S. hospitals about scope-related superbug outbreaks “despicable.”
- 2
Two patients died and six more were sickened in a new outbreak suspected of being caused by contaminated medical scopes, according to a regulatory report by device manufacturer Olympus Corp.
- 3
Soon after doctors at UCLA’s Ronald Reagan Medical Center traced deadly infections to tainted medical scopes last year, they pressed the device maker to lend them replacements.
- 4
After Olympus Corp. paid to fly three doctors from a prominent California hospital to Japan for a weeklong vacation, one of the physicians thanked the company for providing them with “so much extra entertainment that we did not expect.”
- 5
An outbreak at a Pennsylvania hospital in late 2012 should have been an early warning that a reusable medical scope was spreading deadly infections and nearly impossible to disinfect.
- 6
The superbug that raged through Bill Warner’s body after a routine medical scope procedure in early 2013 was so dangerous that his family was warned about entering his room at a time when he needed them most.
- 7
Olympus Corp. said Friday that it would voluntarily recall and redesign a troubled medical scope that has been linked to deadly patient infections around the world.
- 8
Scores of hospital patients treated with medical scopes were infected with potentially deadly bacteria because of repeated failures by manufacturers, regulators and hospitals to report outbreaks, according to a U.S.
- 9
The hunt for a deadly superbug that sickened 22 patients at a Dutch hospital began just before noon on a spring day in 2012.
- 10
Long before the recent superbug outbreaks, Olympus Corp. drew national attention for a faulty device tied to patient infections.
- 11
Recent events involving scope-related outbreaks of antibiotic-resistant superbug infections 2010 August: Japanese device maker Olympus Corp. introduces its TJF-Q180V duodenoscope. 2012 April 23: An Olympus employee dismantles a duodenoscope suspected of infecting 22 patients at a Netherlands hospital.
- 12
A U.S. lawmaker is calling on the Securities and Exchange Commission to investigate Olympus Corp. in response to a Times article that detailed how the company kept selling its scopes despite warnings from a 2012 superbug outbreak in the Netherlands.
- 13
Amid an ongoing investigation into superbug outbreaks nationwide, U.S. regulators have ordered a Pennsylvania company to recall its scope-cleaning machines used at UCLA and more than 1,000 other hospitals and clinics.
- 14
Federal regulators expanded their warnings about medical scopes spreading infections, issuing a new alert Thursday about contaminated bronchoscopes posing a risk to patients.
- 15
Medical scope maker Olympus Corp. said Thursday that it has reached out to a Pasadena hospital that suspects the company’s devices spread a superbug infection to at least three patients.
- 16
A Pasadena hospital is investigating a suspected outbreak related to the same type of medical scope tied to superbug infections across the country.
- 17
- 18
Embattled device maker Olympus Corp. posted a 14% increase in sales of medical scopes worldwide despite health officials linking its instruments to superbug outbreaks at U.S. hospitals.
- 19
In response to deadly superbug outbreaks, the Food and Drug Administration recommended extra steps that hospitals can take to clean medical scopes while work continues on redesigning the troublesome devices.
- 20
A doctor reported in December that a medical scope commonly used to examine patients’ lungs had infected 14 people with a superbug that kills half its victims.
- 21
For decades, doctors reassured patients about the safety of medical scopes with a single statistic.
- 22
Following a superbug outbreak involving three deaths, UCLA officials are planning to test a new scope-cleaning machine designed by a small Arizona company.
- 23
The Justice Department is investigating embattled scope maker Olympus Corp. and its role in recent superbug outbreaks at UCLA and other U.S. hospitals.
- 24
Federal regulators halted manufacturing and sales at a Pennsylvania company in 2012 that made the scope-washing machines used at UCLA and two other hospitals with recent superbug outbreaks.
- 25
Three months after deadly superbug outbreaks sparked alarm nationwide, U.S. hospitals are still searching for how best to clean a controversial medical scope and keep patients safe.
- 26
More than a year before a medical scope infected patients with a superbug at UCLA and Cedars-Sinai medical centers, a similar outbreak in the Netherlands prompted Dutch regulators to ask the manufacturer to prove the device was safe.
- 27
A federal panel concluded Friday that the medical scopes linked to a deadly string of superbug outbreaks are unsafe as designed and urged the U.S.
- 28
Nearly two years before superbug outbreaks hit UCLA and Cedars-Sinai medical centers, the maker of the scopes involved was already warning hospitals in Europe about the risk of such patient infections.
- 29
A Seattle hospital hit by a superbug outbreak is teaming up with a patient’s family and accusing Olympus Corp. of selling a defective medical scope prone to spreading deadly bacteria.
- 30
A federal lawmaker criticized the latest response from the nation’s top health regulator on the recent superbug outbreaks and renewed his push for congressional hearings.
- 31
- 32
Under fire for selling a medical scope linked to superbug outbreaks, Olympus Corp. is pushing back against its critics and says the design of its product isn’t necessarily the sole cause for infections.
- 33
Olympus Corp. and two other manufacturers of medical scopes linked to a string of superbug outbreaks declined to speak Thursday at a Food and Drug Administration hearing on the deadly problem.
- 34
Patients across the nation continue to face considerable risk from medical scopes linked to deadly bacterial outbreaks due to basic design flaws and a lack of easy fixes, a federal panel was warned Thursday.
- 35
Federal regulators shed more light on the potential harm to patients from a controversial medical scope, disclosing 142 reports of contaminated devices and possible patient infections since 2010.
- 36
The cardiac surgeon had unknowingly spread a staph infection from the rash on his hand to the hearts of at least five patients by the time Los Angeles County health investigators learned of the outbreak.
- 37
More patients across the country may have been infected by medical scopes manufactured by Olympus Corp. than previously thought, health officials warned Monday.
- 38
A federal lawmaker is seeking information from Pentax Medical, a manufacturer of medical scopes that have been linked to recent outbreaks of superbug infections.
- 39
Olympus Corp., the maker of medical scopes linked to recent deadly superbug outbreaks, reversed course Thursday and said it was issuing an urgent update on how the device should be cleaned.
- 40
A federal lawmaker said it’s wrong for device-maker Olympus Corp. to profit from recent superbug outbreaks as hospitals buy more of the company’s scopes in response.
- 41
In response to the superbug outbreak at UCLA, the family of a 41-year-old patient who died in January has sued a medical device maker for wrongful death.
- 42
The Food and Drug Administration, under fire for its slow response to superbug outbreaks from tainted medical scopes, said it will now require proof from manufacturers that new devices can be cleaned reliably.
- 43
Joining others in Congress, Sen.
- 44
Well before the recent superbug outbreaks at UCLA and Cedars-Sinai hospitals, federal health officials had labeled deadly CRE bacteria an urgent threat.
- 45
In the latest superbug outbreak, Cedars-Sinai Medical Center discovered that four patients were infected with deadly bacteria from a contaminated medical scope, and 67 other people may have been exposed.
- 46
As more infections come to light from tainted scopes, several members of Congress are demanding answers from the Food and Drug Administration about what it knew about the risk beforehand and for how long.
- 47
The Food and Drug Administration, already under fire for its response to superbug outbreaks at U.S. hospitals, has tried and failed twice to get medical scope manufacturers to prove their controversial devices can be cleaned of deadly bacteria.
- 48
Three years ago, Japanese electronics giant Olympus Corp. was in crisis amid a massive accounting scandal and plunging sales of its signature cameras.
- 49
Cedars-Sinai Medical Center said it’s investigating whether patients with superbug infections are linked to contaminated medical scopes, similar to a recent outbreak at UCLA.
- 50
Following a superbug outbreak at UCLA, the family of a 48-year-old patient who died there has sued a medical device maker for wrongful death.
- 51
A senior Food and Drug Administration official voiced reservations about the new method UCLA’s Ronald Reagan Medical Center is using to clean medical scopes linked to an outbreak of antibiotic-resistant bacteria.
- 52
Prompted by the UCLA superbug outbreak, a federal lawmaker is calling on Congress to investigate what the U.S.
- 53
In the first lawsuit stemming from the superbug outbreak at UCLA, an 18-year-old patient accused a major healthcare device maker of negligence for selling a medical scope prone to spreading deadly bacteria.
- 54
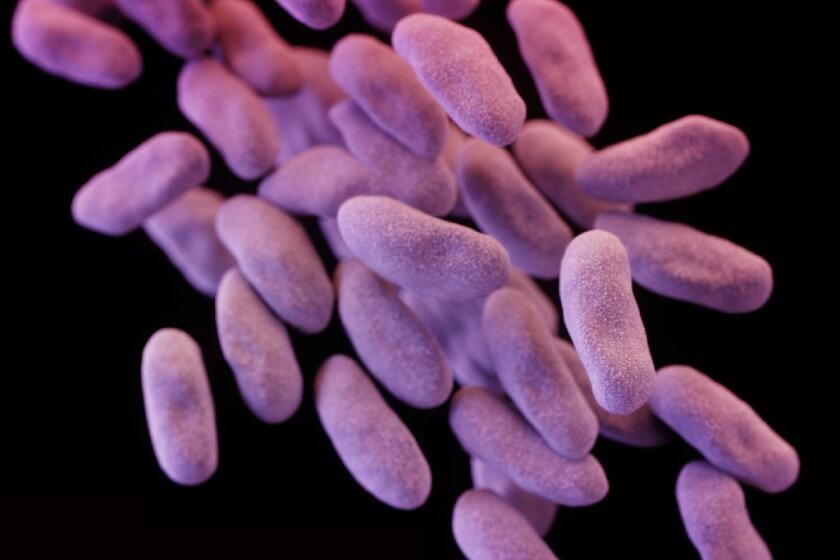
CRE superbugs are becoming more widespread in hospitals and other healthcare facilities.
- 55
Hospitals nationally are scrambling to figure out how to keep using a controversial medical device that benefits patients while avoiding another deadly bacterial outbreak like the one at UCLA Medical Center.
- 56
A commonly used medical scope linked to a deadly bacterial outbreak at UCLA may be so flawed it cannot be properly cleaned, federal officials conceded Thursday.
- 57
Nearly 180 patients at UCLA’s Ronald Reagan Medical Center may have been exposed to potentially deadly bacteria from contaminated medical scopes, and two deaths have already been linked to the outbreak.
- 58
UCLA hospital officials said they began investigating the possibility of a deadly bacterial outbreak in mid-December, but one patient’s account suggests they missed a chance to discover the problem much earlier.
- 59
The manufacturer of the medical scopes at the center of a deadly bacterial outbreak at UCLA Medical Center is under investigation by federal officials for possible violations of laws that ban improper payments to doctors and other customers.
- 60
Consumers might think twice about dining at a restaurant with a poor health grade posted in the window.
- 61
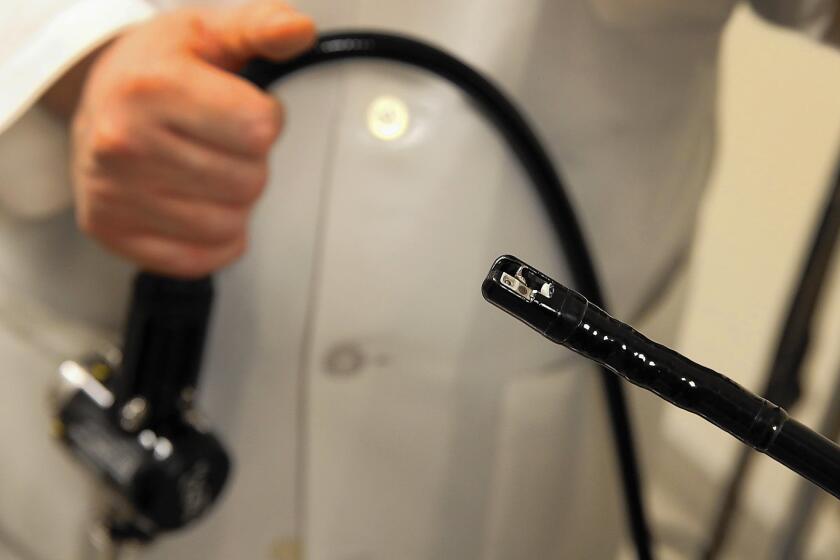
The medical device implicated in the superbug outbreak at UCLA’s Ronald Reagan Medical Center has a reputation for being tough to disinfect, largely due to its unique design, doctors say.